Predicting the Outcome of Critically Ill Children and Adolescents with Electroencephalography
Article information
Abstract
Purpose
Electroencephalography (EEG) is an effective test in predicting severe cortical dysfunction associated with poor outcomes in adult patients, but its value in pediatric patients remains incomplete. Here, we assessed the prognostic value of EEG regarding sedative history and various etiologies in pediatric patients who had undergone EEGs at the pediatric intensive care unit of Severance Hospital for 5 years.
Methods
We performed a retrospective study of total 113 pediatric patients who met our criteria. In-hospital mortality was measured for the primary outcome.
Results
In-hospital mortality was observed in 43 patients (38.1%) and sedatives were used in 37 patients (32.7%). Patients who showed in-hospital mortality were more like to have higher EEG background scores and absent EEG reactivity (P<0.001 for both). The prognostic values of these EEG factors were statistically significant in non-sedated patients (P<0.001 for both) whereas they were not significant in sedated patients (P=0.980 and P=0.336, respectively). In a multivariable regression analysis conducted in non-sedated patients, higher EEG background score and absence of EEG reactivity were independently associated with higher mortality rate (P=0.015 and P=0.001, respectively). They also showed high prognostic values of mortality in non-sedated patients, irrespective of each etiology (hypoxic ischemic encephalopathy [HIE]: P=0.013 and P=0.021, respectively; non-HIE structural brain disease: P=0.001 and P=0.002, respectively; non-structural brain dysfunction: P<0.001 for both).
Conclusion
Our findings prove that both an abnormal background rhythm and the absence of reactivity in early EEG can be independent factors associated with mortality in non-sedated critically ill children irrespective of etiology.
Introduction
Predicting the prognoses of children admitted to pediatric intensive care units (PICUs) for critical care is important to both clinicians and families. Futile investigations and treatments can be avoided when a poor outcome is predicted, while more active management can be pursued with patients whose outcomes are thought to be favorable. Communication with families is also facilitated if poor outcomes are predicted at the start of patient admission to PICU. Therefore, risk factors, laboratory results, and other results from different tests, such as electrocardiography, which are thought to have prognostic values in mortality and morbidity in patients admitted to PICUs have been continuously investigated, and mortality prediction models integrating such factors have been suggested [1,2].
Electroencephalography (EEG), a non-invasive, bedside investigative tool reflecting cortical activities and dysfunction that can be readily applied to both adult and pediatric patients in intensive care units (ICUs), is also well known to have prognostic significance in critically ill patients in several studies [3-6]. EEG is a sensitive tool to detect encephalopathies, and patients with neurologic complications were shown to have an increased risk of mortality compared with patients who do not exhibit these problems [7]. However, previous studies have mostly focused on the prognostic values of EEG in adult ICU patients compared with pediatric patients, and our understanding of specific EEG findings that are associated with poor outcomes in pediatric patients is incomplete. In addition, many of these studies mainly focused on narrow etiologies such as hypoxic ischemic encephalopathy (HIE) or sepsis, rather than diverse etiologies of real ICU situations. Therefore, EEG findings that are associated with poor outcomes in pediatric patients with diverse etiologies admitted to a PICU need to be defined.
Although studies on adults have shown that continuous sedative infusions are associated with prolonged periods of mechanical ventilation and increased mortality, it is difficult to discontinue sedation in pediatric patients who are more vulnerable to self-extubation, adverse cardiovascular effects, ventilator fighting, and negative psychological effects causing frequent sedative use in critically ill children [8-10]. However, background EEG rhythms can be affected by sedatives—increased level of sedation can cause progressive slowing in basic EEG rhythms; fast activities may be more dominant upon light sedation; but increased delta activities, burst-suppression, suppression, and then isoelectric patterns are observed as sedation becomes deeper [11, 12]. Therefore, EEG background rhythms of sedated children may not have prognostic significance because the changes could be the results of sedative effects.
In this context, this study was designed to assess the prognostic value of EEG in children with newly developed neurological disorders of diverse etiologies who were admitted to the PICU, and to discover EEG findings associated with poor outcomes, such as the mortality or morbidity rate. In addition, we compared the EEG findings between the sedated and non-sedated groups to assess the relationship with intravenous sedative infusions.
Materials and Methods
1. Study design and patient selection
A retrospective study was conducted with patients who had undergone EEGs at the PICU of Severance Hospital for 5 years, from January 2012 to December 2016. The inclusion criteria were as follows: (1) patients that had EEGs performed on them within 72 hours of admission to the PICU, and (2) patients who were between the ages of 44 weeks (post-conceptual) and 18 years at the time of admission to PICU. Patients with a history of neurological disorders, epilepsy, developmental delay, or those who had a history of abnormal EEGs, were excluded to avoid complications with baseline abnormalities. Patients admitted to the neurosurgical ICU with a primary diagnosis of head trauma or those that needed neurosurgical intervention were also excluded. This study was approved by the Institutional Review Boards of Severance Hospital (4-2015-0854). We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
2. Clinical variables
In-hospital mortality was selected as a prognostic marker because it could be accurately retrieved from the past chart reviews. Mortality was assessed for each group of patients with the same EEG background scores, and the relationship between mortality and the absence of EEG reactivity was also investigated.
Clinical factors that may be associated with poor outcomes, such as gender, age at EEG recording, duration of ICU or hospital admission, time from ICU admission to EEG initiation, etiologies of ICU admission, reasons for EEG recording, and sedation history, were also collected. Scores of the pediatric risk of mortality III (PRISM III), a mortality prediction model for children admitted to PICUs, measured at the first 24 hours of PICU stay (PRISM III-24), were calculated [1].
The etiologies for PICU admission were categorized into one of the following: (1) HIE diagnosed by computed tomography or magnetic resonance imaging (MRI); (2) non-HIE structural brain disease such as encephalitis or other structural brain damage; (3) functional or non-structural brain dysfunction with no evident brain damage shown on MRI, but with a possible source of brain malfunction such as hypoxic, metabolic, toxic, or infectious etiologies; and (4) others indicating non-central nervous system (CNS) dysfunction [5].
The reasons that led to EEG evaluations in patients were labeled as follows: (1) evaluation after anoxia or hypoxia; (2) alteration in mental status; (3) presence of definite seizures; (4) suspicious movements that are unlikely to be seizures but needed further investigation; and (5) focal neurologic signs.
The patients were then further divided into two groups, one comprising patients who received continuous intravenous infusion of sedatives or analgesics, mostly midazolam, ketamine, fentanyl, or a combination of the three drugs, and the other group consisted of patients that were not sedated at the time when their first EEGs at the PICU were taken.
3. EEGs
Scalp EEGs were performed using gold cup electrodes placed according to the international 10 to 20 system, and they were recorded using a portable video-EEG system (Telefactor, Grass Technologies, Rockland, MA, USA). EEGs were either performed for 30 minutes or monitored continuously (for a minimum of 4 hours) depending on the clinician’s judgment based on ongoing EEG results and the clinical status of the patient. EEG reactivity was checked by eye opening and closing or noise in more alert patients, or by exposure to alerting stimuli such as touch or light pain to more obtunded patients. EEG reactivity was considered present if there was a definite reproducible change in the amplitude and frequency in background activities after exposure to stimuli.
The first EEGs performed after PICU admission within 72 hours for each patient were freshly reviewed by two pediatric neurologists. The EEG backgrounds and presence of reactivity were analyzed. The EEG background scores were recorded according to a scoring system that have shown its usefulness for predicting the neurological prognosis in hypoxic encephalopathy patients, with poorer EEG backgrounds showing higher scores: (1) normal, (2) diffuse slowing (continuous EEG pattern with a dominant frequency less than that appropriate for age: <4 Hz for <6 months of age, <6 Hz for <1 year of age, and <8 Hz for ≥2 years of age), (3) epileptiform (seizures and generalized periodic discharges), (4) burst-suppression (clear increases in amplitude of ≥20 µV [bursts], followed by inter-burst intervals of at least 1 second with low-voltage activity [suppressions]), (5) low-voltage (EEG activity of <20 µV), and (6) isoelectric (without any visible EEG activity) [13]. Next, we divided the background EEG scores into three different groups including two consecutive scores in each group to avoid small-sized patient cohorts. Examples of EEGs from our patients for each background score are demonstrated in Fig. 1. Meetings were held for mutual agreements, but disagreements in background scores and the presence of reactivity did not arise.
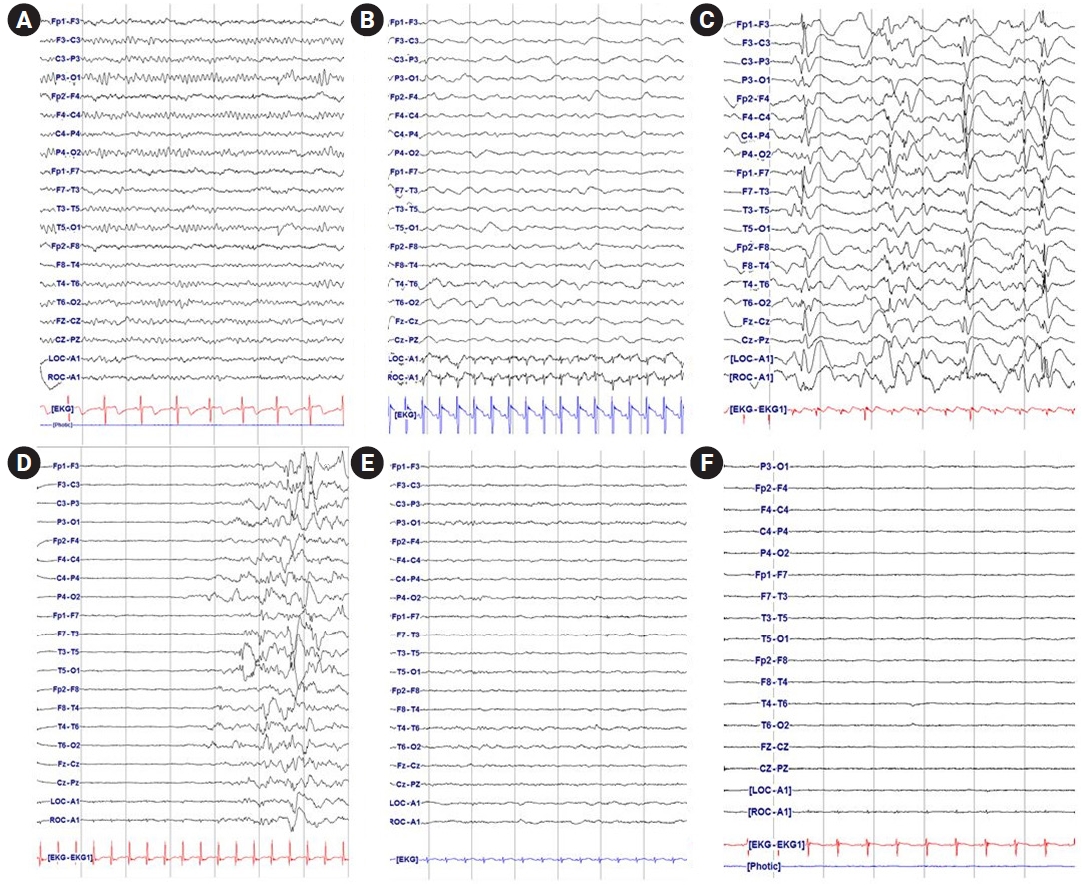
Examples of background electroencephalography (EEG) rhythms of patients in the pediatric intensive care unit. (A) normal background EEG rhythm; (B) diffuse slowing background EEG rhythm (continuous EEG pattern with a dominant frequency less than that appropriate for age); (C) epileptiform (seizures and generalized periodic discharges); (D) burst-suppression (clear increases in amplitude of ≥20 µV, followed by inter-burst intervals of at least 1 second with low-voltage activity); (E) low-voltage (EEG activity of <20 µV); (F) isoelectric (without any visible EEG activity).
4. Statistical analysis
Comparisons of the two groups were performed using chi-square tests or Fisher’s exact tests for categorical or ordinal data and Mann-Whitney U tests for non-parametric continuous data. Univariate regression analysis was also used for risk analysis. To exclude any confounding factors, multivariable regression analysis was conducted. To avoid false-negative results, factors that were highly influenced by each other, such as the background EEG score and absence of EEG reactivity, were analyzed separately. A P<0.05 was considered significant. The SPSS version 23.0 (IBM Co., Armonk, NY, USA) was used for all the analyses. Values were expressed as medians with interquartile ranges (IQRs) for continuous and ordinal variables, or as counts and percentages for categorical variables.
Results
1. Demographics
In total, 221 patients had undergone EEGs at the PICU of Severance Children’s Hospital between January 2012 and December 2016, within 72 hours of admission to the PICU. Of the 221 patients, 108 patients that had a history of neurologic disorder, delayed development, epilepsy before PICU admission, or whose previous EEG results showed abnormal findings were excluded.
The remaining 113 patients were included in this study, and their characteristics are summarized in Table 1. Among them, 65 (57.5%) were boys, and the median age when the first EEGs were performed during the PICU stay was 2.0 years (IQR, 0.5 to 6.2). The median PICU duration was 14 days (IQR, 7 to 32), and the median total hospital duration was 42 days (IQR, 22 to 86). The first EEGs of these patients at the PICU were performed at a median of 19 hours (IQR, 8 to 36) after PICU admission. Thirty-minute standard EEGs were performed in 93 patients (82.8%), and continuous EEG monitoring was performed in 20 patients (17.7%).
Patients with structural brain damage (n=66, 58.4%), including HIE (n=35, 31.0%) and non-HIE structural brain disease, such as encephalitis or other structural brain damage (n=31, 27.4%), accounted for more than 50% of patients who underwent EEG at the PICU. Patients with non-structural brain dysfunction (n=41, 36.3%) also accounted for a large proportion, mainly due to the risk of developing hypoxic encephalopathy from pulmonary diseases (n=17, 15.0%) and developing metabolic encephalopathy due to hepatic failure (n=15, 13.3%). Other than pulmonary and hepatic diseases, renal failure (n=4, 3.5%) and sepsis (n=5, 4.4%) also accounted for some proportion of non-structural brain dysfunction. Six patients (5.3%) were classified as ‘other,’ indicating non-CNS dysfunction.
Among the 113 patients, the most common reason for EEG was a hypoxic or an anoxic event (n=40, 35.4%), followed by alteration of the mental status (n=36, 31.9%), the presence of a seizure (n=20, 17.7%), suspicious seizure-like movements (n=15, 13.3%), and focal neurologic signs (n=2, 1.8%). In addition, sedatives were used before the initiation of EEG recording in 37 patients (32.7%).
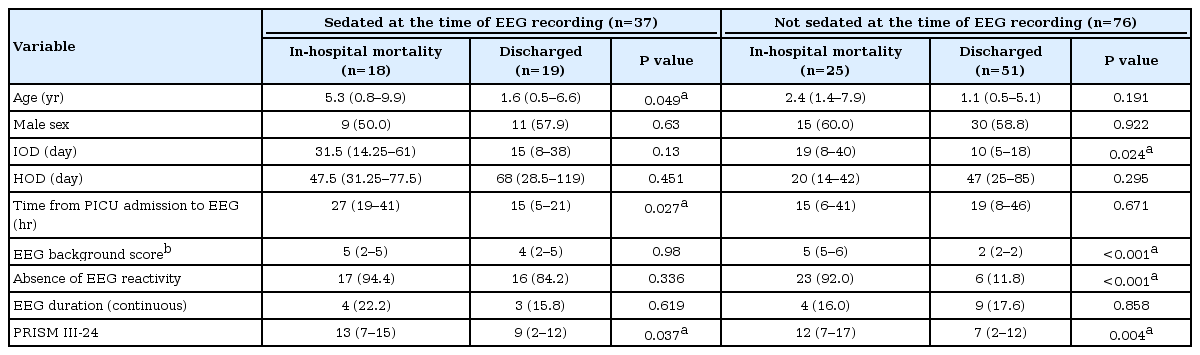
2. Clinical characteristics, EEG background score, and EEG reactivity according to in-hospital mortality
Forty-three patients (38.1%) died during the hospital stay, and 70 patients (61.9%) were discharged from the hospital. Patients who showed in-hospital mortality were significantly older (P=0.012). The length of stay at the PICU was significantly longer in patients who showed in-hospital mortality (P=0.001), and the total length of hospital stay, including periods in the general ward after PICU discharge, was significantly longer in patients that were discharged (P=0.037). The PRISM III-24 score was significantly higher in patients who showed mortality (P<0.001). Both etiologies of PICU admission (P=0.744) and sedation history (P=0.106) were not significantly associated with mortality (Table 1).
The number of patients who showed each EEG background score was as follows: (1) normal (n=12, 10.6%), (2) diffuse slowing (n=45, 39.8%), (3) epileptiform (n=9, 8.0%), (4) burst-suppression (n=4, 3.5%), (5) low-voltage (n=24, 21.2%), and (6) isoelectric (n=19, 16.8%). Among them, the mortality cases were reported as 0, 9, 3, 2, 13, and 16 patients for each EEG background score, respectively, thus showing a higher mortality rate as the background EEG score increased (P<0.001). In addition, of the 62 patients (54.9%) that showed the absence of EEG reactivity, 40 patients showed in-hospital mortality with a significant association between these two variables (P<0.001) (Table 1). When multivariable regression analysis was conducted to exclude confounding clinical factors, the EEG background score (P=0.003) and absence of EEG reactivity (P<0.001) were proven to have prognostic value for in-hospital mortality.
3. Prognostic values of EEG according to sedation at the time of EEG recording
The patients were then further divided into two groups based on receiving intravenous sedative infusion at the time of EEG. Thirty-seven patients (32.7%) were sedated at the time of EEG recording. When the clinical characteristics were analyzed, the length of stay at the PICU was significantly longer in sedated patients (P=0.005). The EEG background score was significantly higher in sedated patients (P=0.012), and the absence of EEG reactivity was significantly higher in sedated patients (P<0.001), thus showing that sedation indeed influences EEG background rhythms and reactivity.
Furthermore, clinical factors that are likely to influence EEG features were analyzed for their association with in-hospital mortality in each group (Table 2). In this univariate analysis, the first EEG was performed significantly late in those who showed in-hospital mortality in the sedated group (P=0.027). In addition, patients with in-hospital mortality were significantly older (P=0.049). However, the EEG background score and absence of EEG reactivity were not associated with in-hospital mortality in the sedated group (P=0.980 and P=0.336, respectively) (Fig. 2A). In the non-sedated group, the duration of PICU stay was significantly higher in patients who showed in-hospital mortality (P=0.024). The EEG background score was significantly higher, and the absence of reactivity was significantly more frequent in patients who showed in-hospital mortality (P<0.001 for both) (Fig. 2B). PRISM III-24 was significantly higher in patients who showed mortality in both the sedated and non-sedated groups (P=0.037 and P=0.004, respectively).
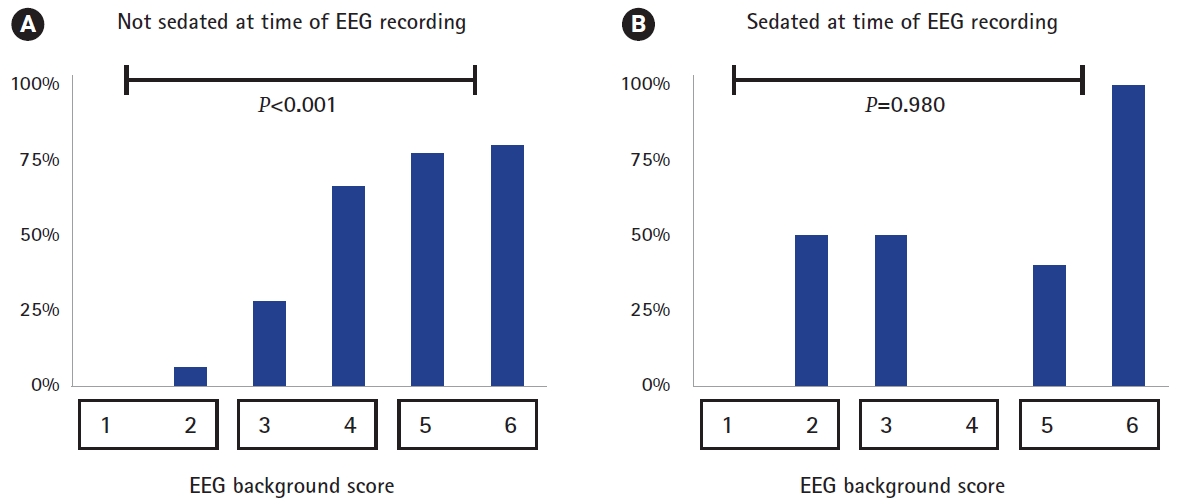
Mortality rate of patients according to sedation at the time of electroencephalography (EEG) recording. (A) Not sedated at time of EEG recording. (B) Sedated at time of EEG recording.
The odds ratio for mortality according to the EEG background score and reactivity adjusted for variables of age at EEG recording, sex, PICU duration, hospital duration, time between PICU admission and EEG initiation, EEG recording duration (standard 30-minute recording vs. continuous monitoring), and PRISM III-24, were then calculated for non-sedated patients at the time of EEG recording (Table 3). In this multivariable analysis, a higher EEG background score showed a significantly higher mortality rate (P=0.015), and the absence of EEG reactivity was also significantly associated with a higher mortality rate (P=0.001).
4. Prognostic values of EEG according to the etiology of PICU admission in non-sedated patients
The prognostic values of EEG were then calculated in non-sedated patients according to the etiologies at PICU admission. In patients diagnosed with HIE, higher EEG background scores (P=0.013) and the absence of EEG reactivity (P=0.021) were highly associated with in-hospital mortality. Higher EEG background scores and the absence of EEG reactivity in patients with other etiologies were also observed, such as non-HIE structural brain disease (P=0.001 and P=0.002, respectively) and non-structural brain dysfunction (P<0.001, both), thus confirming the prognostic value of EEG in various etiologies.
Discussion
The relationship between EEG background rhythms and clinical outcomes has been demonstrated in previous studies, more frequently with cohorts of post-anoxic comatose patients after cardiac arrest. Quasiperiodic generalized spikes on a suppressed background, burst-suppression patterns, low-voltage backgrounds, and isoelectric patterns were each shown to be associated with mortality and poor neurologic outcome in post-anoxic comatose adult patients after cardiac arrest [4,14-17]. In addition, in a study with sedated or comatose adults in ICU with various underlying diseases, a burst-suppression pattern was associated with increased mortality; in another study with septic adult ICU patients, diffuse slowing, burst-suppression, and an isoelectric pattern were associated with mortality [3,4,18,19]. In children aged older than a post-conceptual age of 44 weeks, the available data are scant, but several studies have shown similar results to those of adult studies: epileptiform backgrounds, burst-suppression patterns, low-voltage backgrounds, and isoelectric backgrounds were associated with mortality and poor neurological outcomes in patients with HIE after cardiac arrest [20-23]. In children receiving critical care for reasons other than HIE, low-voltage, and isoelectric backgrounds were associated with poor outcomes in children with non-traumatic coma, diffuse slowing, epileptiform, and suppressed backgrounds were associated with mortality in children with acute liver failure, and diffuse slowing background was associated with poor outcomes in near-drowning encephalopathy patients [24-27].
Previous studies largely show the association between the absence of reactivity and increased mortality and morbidity in adult patients [3,4,19,21,28,29]. Although fewer studies were conducted in children, a study by Ramachandrannair et al. [30] showed an association between the absence of EEG reactivity and increased mortality and neurologic impairment in comatose children.
In our study, a higher EEG background score and the absence of EEG reactivity from EEGs performed within 72 hours from PICU admission were associated with an increased risk of mortality after adjusting for clinical factors in non-sedated patients, a finding that is concordant with previous study findings. No mortality was found with a normal EEG; in addition, compared with patients with a normal EEG or with only diffuse slowing, patients whose EEG showed epileptiform backgrounds or burst-suppression patterns were 193.3 times more likely to die (95% confidence interval [CI], 3.4 to 10,886.1; P=0.010), and patients with low-voltage or isoelectric EEG were 692.6 times more likely to die (95% CI, 7.6 to 62,708.2; P=0.004), respectively. Patients with absent EEG reactivity were 234.1 times more likely to die than patients who showed EEG reactivity (95% CI, 9.4 to 5,831.9; P=0.001) (Table 2).
Changes in the EEG background rhythm and EEG reactivity with an increasing level of sedation were also demonstrated in previous reports [12,31]. In our study, sedation alone significantly influenced the EEG background and reactivity, and the EEG background score and reactivity did not predict mortality if EEG was performed in sedated patients but predicted mortality in non-sedated patients. However, in a study by Azabou et al. [4] conducted with septic adult patients in the ICU, excessive sedation was not related to the absence of EEG reactivity [11]. This discrepancy is probably due to the deeper level of sedation observed in our patients with a median Richmond agitation-sedation scale (RASS) of –4 (IQR, –3 to –4) compared with –2 in the study by Azabou et al. [4]. A deeper level of sedation is possibly associated with EEG background change and the absence of EEG reactivity [32]. Therefore, although EEG findings can be used as a prognostic factor in children in the PICU, accurate prediction of outcomes in deeply sedated patients at the time of recording may be difficult.
EEGs in patients with HIE, which often accompanies neuronal deaths, have been demonstrated to be more useful in predicting prognoses compared with those in patients receiving critical care with other etiologies [3]. There is limited literature on the relationship between the EEG background and clinical outcome in critically ill patients with etiologies other than HIE, and even fewer in children in the PICU. However, neurologic complications commonly occur in critically ill children without a definite history of anoxia. In addition, in our study comprising patients admitted to the PICU with various reasons, the EEG background rhythm showed an association with mortality, suggesting that EEG is more widely applicable to diverse patient populations.
This study is significant because it is the first study to our knowledge, to investigate the relationship between early EEG findings and mortality outcomes in critically ill children with diverse etiologies, irrespective of the comatose status, as well as the association with sedatives.
However, this study has several limitations. Due to its retrospective nature, the depth of sedation at the exact time of EEG recording was not retrieved. Instead, the RASS scores charted every 8 hours closest to the time of EEG recording were collected. Therefore, changes in the EEG background rhythm and reactivity according to the depth of sedation could not be assessed. In addition, because 82.3% of patients underwent standard 30-minute EEG while only 17.7% of patients were studied with continuous EEG monitoring, seizure burdens that could affect the prognosis in critically ill children could not have been assessed [33]. The trends and changes in the EEG background rhythms over time is also another prognostic factor, but this also could not be analyzed [34,35]. Finally, univariate and multivariable regression analysis could not be performed in non-sedated patients with each different etiologies because of the small-sized cohort.
In conclusion, abnormal background rhythms and absent reactivity in early EEG can be an independent predictor for mortality in non-sedated critically ill children in the PICU, irrespective of the etiology.
Notes
No potential conflicts of interest relevant to this article was reported.
Acknowledgements
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare, Republic of Korea (grant number: HI15C1601).