A Case of Progressive Multifocal Leukoencephalopathy in a Child with Hyper-Immunoglobulin M Syndrome: The Impact of Missed Care during the COVID-19 Pandemic
Article information
Progressive multifocal leukoencephalopathy (PML) is a frequently fatal subacute demyelinating disease of cerebral white matter caused by the human polyomavirus 2, commonly known as the John Cunningham virus (JCV) [1]. PML is primarily reported in patients with severe immunosuppression caused by human immunodeficiency virus (HIV) infection, hematologic malignancy, or immunosuppressive therapy, including natalizumab for multiple sclerosis and rituximab for Crohn’s disease [1]. However, PML has also been reported in primary immunodeficiencies (PID), including those with hyper-immunoglobulin M syndrome (HIGM), common variable immunodeficiency, and Wiskott-Aldrich syndrome [2]. In this case study, we describe PML in an immunocompromised child with HIGM during the coronavirus disease 2019 (COVID-19) pandemic.
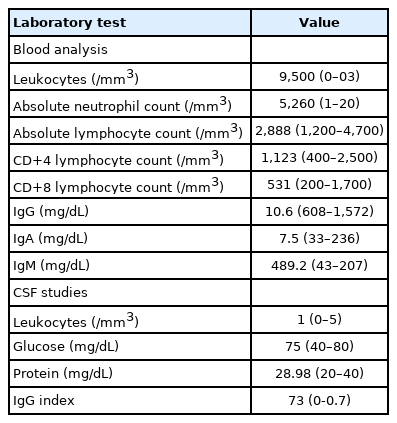
A 6-year-old boy with a history of HIGM was examined after experiencing 3 weeks of left-side weakness in June 2021. The patient had a history of recurrent otitis media and pneumonia since 12 months of age and pertussis at 30 months of age. The patient was diagnosed with HIGM after the pertussis workup. The patient had no family history of immunodeficiency, and his development was normal. The patient had been treated with monthly intravenous immunoglobulin (IVIG) replacement therapy, which had been discontinued 7 months previously because the patient’s parents thought it would be risky to visit a hospital during the COVID-19 pandemic and underestimated the risk of opportunistic infections. The patient had been showing fatigue and poor concentration for 3 weeks prior to the visit and decreased left hand and arm movement for 10 days prior to the visit. The patient did not show signs of upper respiratory or gastrointestinal infection symptoms. The patient was mentally alert; however, a physical examination revealed additional central left facial palsy, urinary incontinence, mutism, and cognitive decline. The patient’s motor grade of the left upper extremity was rated as grade III, and the left lower extremity was rated as grade IV. Right upper and lower motor function was intact. No pathologic reflexes were found. Serum inflammatory markers, including C-reactive protein and the erythrocyte sedimentation rate, were within the normal range. Lymphocyte subset counts were also within the normal range for the patient’s age (Table 1). Serum immunoglobulin (Ig) G and IgA levels were extremely low, with elevated IgM levels (Table 1). Serum HIV antigen and anti-HIV antibodies were not detected. Antibodies against serum anti-myelin oligodendrocyte glycoprotein were absent. The cerebrospinal fluid (CSF) analysis was unremarkable (Table 1). CSF herpes simplex virus polymerase chain reaction (PCR), enterovirus PCR, and oligoclonal band were negative, and no bacteria were grown on the CSF aerobic culture. No acid-fast bacilli or fungi were grown on the CSF culture, and no encapsulated yeast was observed on direct fungal microscopy. Brain magnetic resonance imaging (MRI) with contrast enhancement demonstrated bilateral asymmetric multifocal regions at the subcortical white matter with high signals on the T2-weighted images (Fig. 1). IVIG replacement therapy was initiated due to the low levels of IgG. Although PML was suspected based on the MRI findings and the history of the patient’s immunodeficiency, intravenous methylprednisolone pulse therapy was initiated before other encephalopathies including acute disseminated encephalomyelitis were excluded. JCV PCR tests of the CSF and urine were positive, and the patient was diagnosed with PML. The patient showed partial improvement of facial palsy and lower extremity weakness with slight or no improvement of hand strength and was discharged after 8 days in the hospital. Three weeks after the patient was first admitted, he was readmitted due to worsening of his left-side weakness and an inability to ambulate. The patient’s right hand and leg presented involuntary movements, and the patient could not speak and showed little voluntary movement, such as nodding. Follow-up MRI with contrast enhancement demonstrated worsening of the PML lesions (Fig. 1). Despite 10 additional hospital admissions with periodic IVIG replacement therapy, the disease worsened. Currently, 11 months after the original onset of the disease, the patient is bedridden with a tracheostomy, home ventilation care, and percutaneous intestinal gastrostomy.
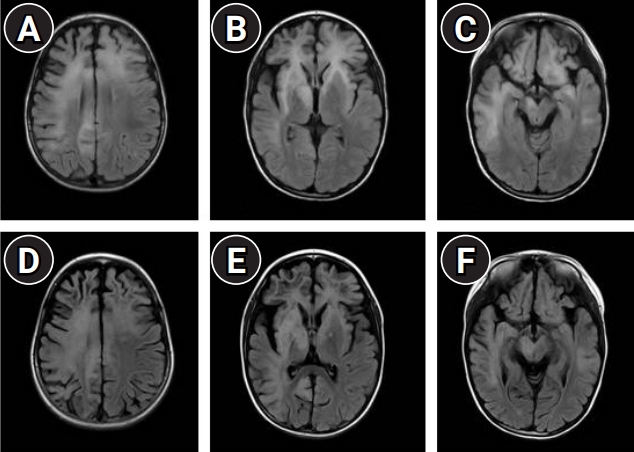
Brain MRI T2 fluid-attenuated inversion recovery (FLAIR) of the patient. Initial brain MRI shows multifocal legions with a high signal on T2 and mostly on the white matter (A, B, C). Follow-up MRI shows worsening lesions with a spongy-like appearance and diffuse cerebral atrophy (D, E, F).
X-linked HIGM, which is the most common type of HIGM, is caused by pathogenic variations in the CD40LG gene. A genetic analysis revealed a 4,858-base pair deletion including exon 5 on the CD40LG gene, which encodes CD40 ligand, in this patient. Patients with HIGM syndrome have extremely low levels of serum IgG due to a class-switching recombination defect. Therefore, regular IVIG replacement therapy is important even if it cannot fully prevent opportunistic infections. Unfortunately, this patient did not receive IVIG replacement during the COVID-19 pandemic. In addition, the CD4+ lymphocyte count is not the appropriate indicator of cellular immunity in HIGM patients because T cell counts are not affected by defects in CD40 ligand expression; instead, CD40 ligand mediates T cell interactions with monocytes, macrophages, and dendritic cells [3]. Therefore, an early classification of the type of HIGM via genetic testing or a functional assessment using flow cytometry is important to decide the appropriate prophylactic or corrective treatment. Hematopoietic stem cell transplantation is the mainstay corrective therapy for HIGM patients with CD40 ligand dysfunction [3]. However, transplant-related complications are also life-threatening, so careful consideration should be given to whom and when stem cell transplantation is performed [4].
The incidence of PML was estimated to be 44 cases per 10,000,000 individuals from 2002 to 2004 in the United States [5]. Zerbe et al. [2] found 26 reported cases of PML with PIDs. Four of those cases were diagnosed with HIGM. After the publication of Zerbe et al. [2], two additional PML cases with HIGM were reported [6]. This case is the first description of PML associated with HIGM in Korea, to the best of our knowledge. As any area of the brain can be involved, the clinical presentations of PML vary. Common clinical presentations include insidious cognitive dysfunction, aphasia, motor or sensory dysfunction, and coordination and gait difficulties [1,7,8]. Brain MRI of PML typically shows multiple hyperintense lesions on T2-weighted images and the fluid-attenuated inversion recovery sequence in the affected white matter with or without gadolinium enhancement. PML is commonly seen as multifocal lesions in the subcortical white matter of the frontal and parieto-occipital lobes. However, PML involvement is possible anywhere in the brain, and it can be seen as an isolated lesion [8]. The histopathologic features of brain biopsies with PML show a triad of multifocal demyelination, enlarged bizarre astrocytes, and enlarged oligodendrocyte nuclei. A definitive diagnosis of PML requires typical histological findings with evidence of JCV infection in the central nervous system. However, brain MRI features and a clinical presentation consistent with PML with positive JCV PCR in the CSF are also sufficient to diagnose PML, as in this patient [8].
The main goal of PML treatment is immune reconstitution. The introduction of antiretroviral therapy significantly improved the outcomes of HIV-related PML patients. In immunomodulatory drug-induced PML, the discontinuation of these drugs can be considered to improve the patient’s outcome [7]. Therefore, a poor prognosis is anticipated when immune reconstitution is not feasible [7]. In the past few decades, several antiviral therapies, including cidofovir for treating JCV, have been investigated with poor results [1]. There was no decrease in mortality or improvements in disability with cidofovir treatment for HIV-related PML [9]. Cytarabine seemed promising due to its ability to inhibit JCV replication in vitro. Nevertheless, several clinical trials could not show decreased mortality in PML patients treated with cytarabine [1]. JCV-specific therapy to improve cellular immunity, including anti-programmed death-1 (anti-PD-1) antibodies (nivolumab and pembrolizumab) and interleukins, have also been tested over the past few years [7]. However, evaluations are limited to case reports or case series. Further studies are needed to evaluate the exact contributions of these therapies to PML treatment. In this case report, IVIG replacement therapy was inadequate for reconstitution of cellular immunity in this patient, which may indicate a poor prognosis. Therefore, hematopoietic stem cell transplantation should be considered in patients diagnosed with PID with CD40 ligand deficiency [4]. Although immune reconstitution is the mainstay of treatment of PML, it paradoxically can induce PML-immune reconstitution inflammatory syndrome (IRIS), which presents as acute clinical worsening due to the hyperinflammation at the site of the original PML lesions [10]. Contrast enhancement and edema of previous PML lesions in MRI are characteristic of this inflammation, which is not a usual sign in classic PML lesions [1]. It has been shown that steroid therapy can be helpful in PML-IRIS patients [10]. We tried steroid treatment, considering the possibility that this patient may have had another steroid-responsive disease, such as acute disseminated encephalomyelitis, not PML. Another reason for using steroids was to prevent inflammation caused by acute immune reconstitution resulting from IVIG.
Herein, we report a case of PML in a boy with HIGM who discontinued treatment during the COVID-19 pandemic. Although cases of PML are relatively rare in PID and largely reported in adult patients with acquired immunodeficiency, PML should be suspected when a patient with PID shows subacute neurologic symptoms with multifocal brain lesions primarily in the white matter.
The present study was approved by the Institutional Review Board of Hallym University Health System (IRB No. 2022-05-017). The board waived the requirement of informed consent.
Notes
There is no financial conflict with GENOME INSIGHT Inc.
Author contribution
Conceptualization: HWK and HJY. Data curation: AYP, HWK, and HJY. Formal analysis: HWK and HJY. Genetic data analysis: SL. Project administration: HJY. Visualization: AYP and HJY. Writing-original draft: AYP. Writing-review & editing: HWK, SL, and HJY.