 |
 |
- Search
| Ann Child Neurol > Volume 30(1); 2022 > Article |
|
Sjögren syndrome is a chronic autoimmune inflammatory disorder that classically presents with diminished lacrimal and salivary gland function, resulting in xerophthalmia and xerostomia [1]. Patients often present with the classic sicca symptoms and are usually diagnosed after 40 years of age. Sjögren syndrome in children is very uncommon. Unlike in the adult population, the sicca symptoms have a low sensitivity in the diagnosis of pediatric Sjögren syndrome since patients are rarely symptomatic at the time of presentation [2]. The spectrum of clinical symptoms is diverse, and extra-glandular symptoms such as musculoskeletal, neurological, renal, and dermatologic involvement have been described in primary Sjögren syndrome. In the literature, neurological manifestations have been reported with variable frequency (from 8.5% to 70%) of patients with primary Sjögren syndrome, and these manifestations frequently preceded the classic sicca symptoms [2-4]. The neurological manifestations of Sjögren syndrome most commonly involve peripheral neuropathy, predominantly in the form of sensory neuropathy [1]. Motor impairment has not been widely recognized in patients with Sjögren syndrome, especially in pediatric cases. To the best of our knowledge, the following case is the first description of a rare presentation of an adolescent patient with primary Sjögren syndrome presenting with isolated limb weakness.
A 15-year-old otherwise healthy male presented to our hospital with sudden-onset bilateral lower limb weakness that started a week prior to the visit. He was able to stand still but had difficulty walking without support. He had experienced mild fever and diarrhea for 2 days around the time of the onset of limb weakness, but exhibited no accompanying symptoms including fever, parotitis, or lymphadenopathy at presentation. He denied any major traumatic event before the development of the symptom. There was no family history of autoimmune disorders. His physical examination revealed symmetric weakening of the lower extremities with an initial motor strength of grade 4/4 without any sensory dysfunction. Neurological examinations showed well-preserved deep tendon reflexes and intact cerebellar function. The initial laboratory workup showed no notably abnormal results with a normal complete blood count, C-reactive protein, and erythrocyte sedimentation rate (Table 1). An examination of the cerebrospinal fluid was unremarkable and showed no pleocytosis, but a normal concentration of glucose and protein. Albuminocytologic dissociation and oligoclonal bands were all absent, and viral studies and cultures for bacterial pathogens were all negative (Table 1). The urinalysis was also unremarkable, but multiplex real-time polymerase chain reaction of a stool sample tested positive for Clostridium perfringens. Magnetic resonance imaging (MRI) of the spine was obtained on day 2 of admission and revealed contrast enhancement around the peripheral nerve roots in the sacral canal. Given this MRI finding, Guillain-Barré syndrome was initially considered as one of the most likely diagnoses, and intravenous immunoglobulin (2 g/kg over 5 days) was administered. However, a nerve conduction study showed slow conduction velocities on the right superficial peroneal sensory nerve and did not show any motor nerve demyelination consistent with Guillain-Barré syndrome. Further, the patient also had a poor response to immunoglobulin therapy.
Based on the results, we considered that an autoimmune or infectious etiology may have been responsible for the patient’s neurological symptom. Serological laboratory tests were then obtained to evaluate possible autoimmune diseases, and the results showed positivity for anti-SSA (Ro) antibody elevated to 28.5 AU/mL (normal <12). Other laboratory findings, including antiganglioside antibodies and antinuclear antibodies (ANAs), were all normal or negative (Table 1).
Although rare, juvenile Sjögren syndrome was suspected due to anti-SSA (Ro) antibody positivity. To further evaluate the possibility of Sjögren syndrome, the patient was referred to ophthalmology and noted to have a normal Shirmer test. However, upon extensive questioning for the review of systems, he was found to have some degree of xerophthalmia characterized as occasional discomfort from dry eyes. He was then referred to rheumatology, and a salivary gland scan showed mildly decreased baseline uptake of radiotracer and markedly decreased excretory function of the bilateral parotid glands (Fig. 1). According to the 2016 American College of Rheumatology/European League Against Rheumatism (ACR-EULAR) classification criteria for primary Sjögren syndrome and known pediatric considerations, we diagnosed the patient as having juvenile Sjögren syndrome based on the evidence of significantly decreased salivary gland function from the salivary gland scan and anti-SSA (Ro) antibody positivity, together with the subjective complaint of intermittent dry eyes [5]. While other rheumatologic etiologies such as systemic lupus erythematosus were considered in the differential diagnosis, the clinical symptoms or serological markers of the patient were not consistent with other diseases.
A 3-day course of pulse methylprednisolone (a total of 3,000 mg) was initiated, followed by oral prednisolone, hydroxychloroquine (300 mg/day), and an immunosuppressive agent, azathioprine (50 mg/day). A 3-week follow-up MRI examination showed a slight improvement in contrast enhancement around the ventral nerve roots in the sacral canal, but the subjective complaint of limb weakness still remained. The patient was discharged on these medications and will receive routine follow-up to monitor any changes in symptoms and prevent further sequelae of the disease.
Sjögren syndrome is a heterogenous autoimmune inflammatory disease usually diagnosed in the adult population. Pediatric Sjögren syndrome was first reported in the 1960s but is generally considered to be a rare, poorly defined, and possibly underdiagnosed condition due to the lack of typical sicca symptoms at presentation [6]. Prior case series and systematic reviews have demonstrated that parotitis is the predominant manifestation of the disease in the pediatric population [7]. The clinical manifestations of pediatric Sjögren syndrome, however, may vary more than those seen in adult patients with involvement of extra-glandular symptoms [6]. In the literature, neurological manifestations of Sjögren syndrome have been reported in up to 70% of patients with primary Sjögren syndrome [3]. Choe et al. [8] reported a 12-year-old girl diagnosed with neuropsychiatric systemic lupus erythematosus associated with secondary Sjögren syndrome presenting with limb weakness and altered mental status, which was a rare case of neurological complications of pediatric Sjögren syndrome. However, to the authors’ knowledge, primary Sjögren syndrome presenting with isolated motor dysfunction as limb weakness in a pediatric patient has not yet been reported in the literature, and the incidence of limb weakness in Sjögren syndrome is unknown in children. Our report is the first description of primary Sjögren syndrome presenting with isolated limb weakness in a pediatric patient.
In the literature, 89% of patients who initially presented with peripheral neuropathy with limb weakness were diagnosed with conditions such as Guillain-Barré syndrome and chronic inflammatory demyelinating polyneuropathy prior to being diagnosed with Sjögren syndrome, resulting in a delay of appropriate treatment [1]. Due to the rarity of juvenile Sjögren syndrome and the unspecific symptoms present in our patient, along with the suspicious MRI findings, we also suspected Guillain-Barré syndrome as the initial diagnosis. However, since no definite motor neuropathy or demyelination was evident in the nerve conduction study of our patient, we presumed that the enhancement of the nerve roots in the sacral canal observed on MRI may have been caused by radiculitis, an inflammation of the nervous system that occurs as a complication of Sjögren syndrome. Furthermore, it is believed that multiple environmental factors such as viral infection may trigger Sjögren syndrome in genetically predisposed individuals; however, infection due to C. perfringens as a cause of Sjögren syndrome has never been reported in the literature.
Although the precise pathogenesis remains unknown, neurological symptoms such as limb weakness may be associated with Sjögren syndrome, and it is important to consider screening for Sjögren syndrome in a child who presents with a new-onset isolated limb weakness even in the absence of definite sicca symptoms to provide appropriate, timely treatment and prevent disease progression.
One of our limitations was that salivary gland biopsy, which is a gold standard in the diagnosis of Sjögren syndrome in the adult population according to the 2016 ACR-EULAR classification, and usually demonstrates the disease as lymphocytic infiltration, was not performed due to the invasiveness of the biopsy procedure. Instead, we performed a salivary scan which confirmed decreased excretory function of both parotid glands.
There is currently no official consensus on the classification criteria of pediatric Sjögren syndrome. The official diagnostic criteria used in adults are often not applicable to pediatric-onset Sjögren syndrome, and the diagnostic tools used in adults may not be the most appropriate evaluation method for children. In order to better understand and further characterize pediatric Sjögren syndrome, it is necessary to establish highly sensitive diagnostic criteria specific for pediatric patients and to have a high index of suspicion of the disease when a child presents with neurological symptoms such as limb weakness, even in the absence of the sicca symptoms, to facilitate an earlier diagnosis.
This study was approved by the Institutional Review Board of CHA Bundang Medical Center (IRB No: 2021-04-017-001). The requirement for written informed consent from the patient was waived by the board due to the retrospective nature of the study.
Notes
Author contribution
Conceptualization: SJB, THH, and KYC. Data curation: SJB, THH, and KYC. Formal analysis: SJB, SYJ, and JIB. Project administration: SJB and KYC. Visualization: SJB and JIB. Writing-original draft: SJB. Writing-review & editing: SJB and KYC.
Fig. 1.
Salivary gland scan of the patient. (A) Salivary scan is performed starting immediately after a bolus intravenous injection of 370 MBq 99mTc-sodium pertechnetate (1 minute per frame). Imaging continued for 30 minutes after injection. Lemon juice is administered orally at 20 minutes after injection and the patient is instructed to swallow the lemon juice at 26 minutes after injection. (B) Purple and blue; parotid glands, yellow and red; submandibular glands, green; oral cavity. (C) Significantly delayed salivary excretion from both parotid glands compared to that from submandibular glands after stimulation of salivary excretion at 20 minutes is shown by the graph.
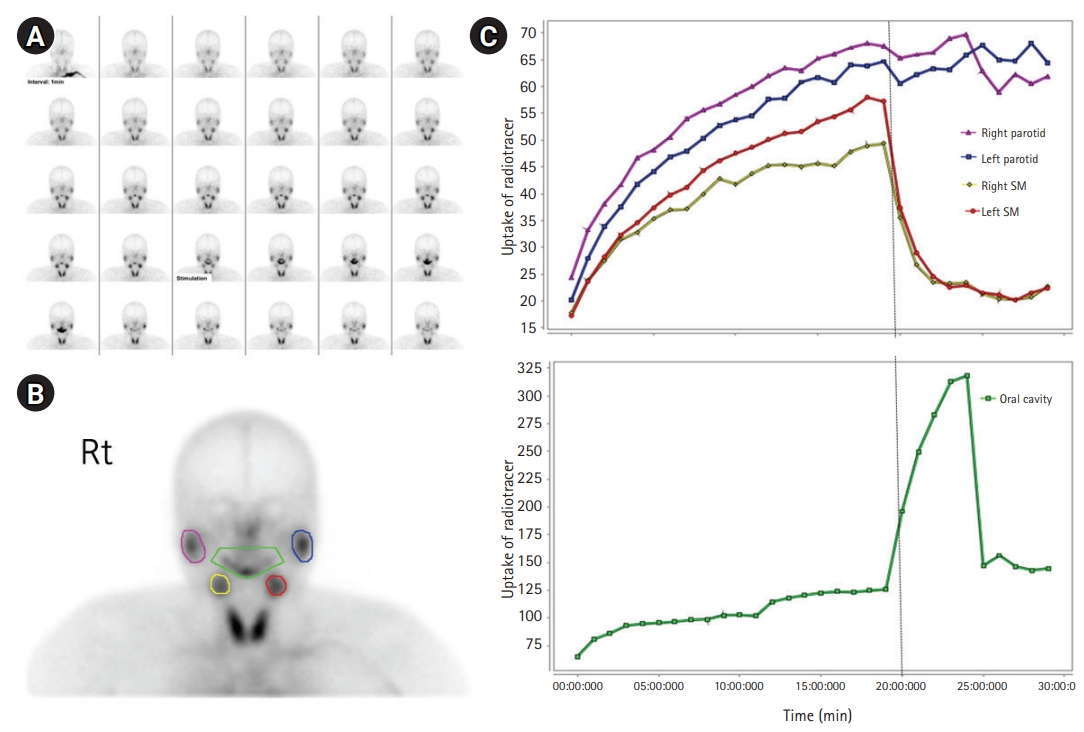
Table 1.
Findings of the serum and cerebrospinal fluid evaluations
| Laboratory test | Result (reference range) |
|---|---|
| Blood analysis | |
| C-reactive protein (mg/dL) | 0.11 (0-03) |
| Erythrocyte sedimentation rate (mm/hr) | 2 (1-20) |
| Creatine kinase (U/L) | 93 (0-190) |
| 25-(OH) vitamin D (ng/mL) | 23.66 (30-100) |
| Thyroid function test | Normal |
| Gangliosides IgM panel | All negative |
| Acetylcholine receptor Ab (nmol/L) | <0.02 (<0.5) |
| ANA | Negative |
| Rheumatoid factor (IU/mL) | <10 (<14) |
| SS-A/Ro Ab (AU/mL) | 28.5a (<18) |
| SS-B/La Ab (AU/mL) | <3.0 (<18) |
| Sm-Ab (anti Smith Ab) (AU/mL) | 14.2 (<18) |
| Anti-dsDNA Ab IgG (IU/mL) | <10.0 (<30) |
| CSF studies | |
| Leukocytes (/mm3) | 1 (0-5) |
| Glucose (mg/dL) | 58 (40-80) |
| Protein (mg/dL) | 32 (15-45) |
| IgG index (mg/dL) | 2.7 (0-4) |
| Oligoclonal bands | Negative |
| Bacterial culture | Negative |
References
1. Seeliger T, Prenzler NK, Gingele S, Seeliger B, Korner S, Thiele T, et al. Neuro-Sjogren: peripheral neuropathy with limb weakness in Sjogren’s syndrome. Front Immunol 2019;10:1600.



2. Lin J, Gadde JA, Flanagan E, Gombolay G. A presentation of pediatric Sjogren’s syndrome with abducens nerve palsy. Neuropediatrics 2021;52:394-7.


3. Perzynska-Mazan J, Maslinska M, Gasik R. Neurological manifestations of primary Sjogren’s syndrome. Reumatologia 2018;56:99-105.



4. Gono T, Kawaguchi Y, Katsumata Y, Takagi K, Tochimoto A, Baba S, et al. Clinical manifestations of neurological involvement in primary Sjogren’s syndrome. Clin Rheumatol 2011;30:485-90.


5. Shiboski CH, Shiboski SC, Seror R, Criswell LA, Labetoulle M, Lietman TM, et al. 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjogren’s syndrome: a consensus and data-driven methodology involving three international patient cohorts. Arthritis Rheumatol 2017;69:35-45.


6. Yokogawa N, Lieberman SM, Sherry DD, Vivino FB. Features of childhood Sjogren’s syndrome in comparison to adult Sjogren’s syndrome: considerations in establishing child-specific diagnostic criteria. Clin Exp Rheumatol 2016;34:343-51.

- TOOLS
-
METRICS

-
- 0 Crossref
- Scopus
- 3,207 View
- 78 Download
- Related articles in Ann Child Neurol
-
A Case of Walker-Warburg Syndrome Presented with Seizures.2010 November;18(2)
A Case of Idiopathic Long QT Syndrome Presenting as Epilepsy.1998 May;6(2)






