Aseptic Zoster Meningoencephalitis with Ophthalmic Shingles in an Immunocompetent 3-Year-Old Boy
Article information
Zoster is uncommon in immunocompetent children, especially those younger than 10 years. It is caused by reactivation of the varicella-zoster virus (VZV), which remains dormant in the sensory ganglia following primary infection with varicella or vaccination. Zoster may involve various neurological complications, such as meningitis, encephalitis, myelitis, meningoencephalitis, cranial neuropathy, and peripheral neuropathy. Here, we present a case of aseptic zoster meningoencephalitis with ophthalmic shingles in a young immunocompetent toddler.
A previously healthy 3-year-old boy was admitted because of progressive irritability and headache. He had hit his right forehead against a door handle 4 days before admission. He presented with a headache, painful erythematous swelling, and ptosis in the right periorbital area. In the emergency room, his vital signs were stable. He was clearly conscious and responded to verbal stimuli. The pupils were symmetrically reactive to light. However, he could not open his eyes and refused to stand up. His initial Glasgow Coma Scale score was 12 (E1, V5, M6). He had no neck stiffness, and all meningeal irritation signs were negative. Initial laboratory tests for blood and cerebrospinal fluid (CSF), as well as neuroimaging studies, were unremarkable.
Aseptic zoster meningoencephalitis was suspected after the patient showed a painful vesicular skin eruption on the right forehead with progressive lethargy on the second day of hospitalization (Fig. 1A). His diagnosis was confirmed by a positive VZV polymerase chain reaction (PCR) test result from a CSF sample on day 4. The CSF culture showed no bacterial growth. Serum varicella immunoglobulin M (IgM) and varicella PCR results were negative; however, the varicella IgG result was positive (3.1; reference < 0.9). The patient had been vaccinated for varicella at 1 year of age. Normal results were found for laboratory tests for immunodeficiency, including the lymphocyte subset, immunoglobulin levels, and complements, except decreased CD4 positive helper T cells (Table 1). A human immunodeficiency virus(HIV) antigen test result was negative. After confirming the diagnosis, intravenous acyclovir was administered for 24 days until the CSF VZV results turned negative. The patient fully recovered and was discharged without further neurological complications.
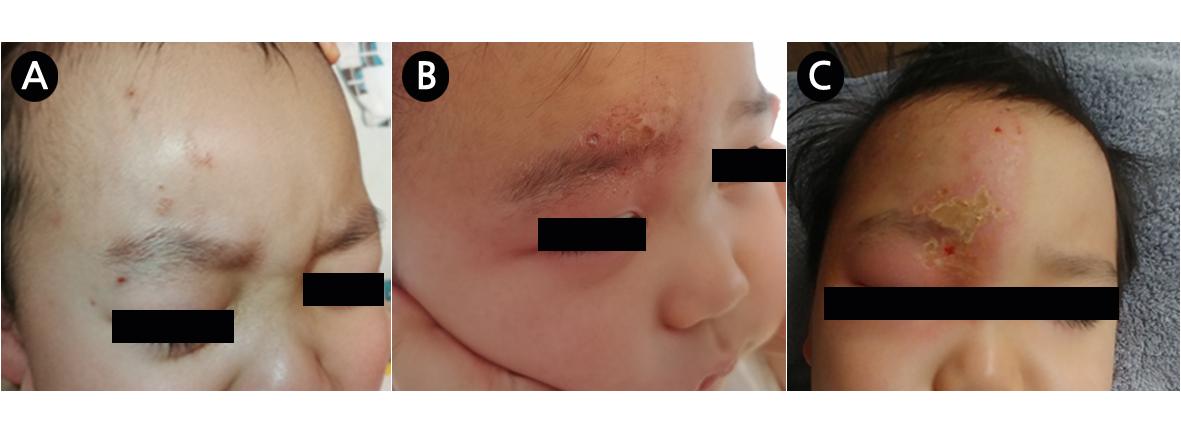
Maculopapular rash evolving into vesicles with erythematous regions on the right V1 dermatome. (A) Hospital day (HD) 2, (B) HD 7, (C) HD 9.
In this case, an early diagnosis was challenging because of the patient’s history of forehead trauma, the late onset of dermatological manifestations of zoster, the absence of pleocytosis on CSF examination, and especially the patient’s young age. Immunocompetent and immunocompromised children showed a similar distribution of age at herpes zoster onset. Pediatric herpes zoster is very common between 10 to 14 years of age [1]. The incidence of zoster in children peaks twice: at 4–5 and 10–13 years of age [2]. Several cases of pediatric herpes zoster meningitis have been reported in Korea, all of which were in children between 8 and 14 years of age, with no cases observed in children under 5 years of age [3,4]. Earlier reports have stated that zoster occurs at a significantly shorter interval if VZV infection is acquired during infancy [5]. Although our patient had been exposed to chickenpox at 70 days of age, he had never developed a clinically evident varicella infection. His early VZV exposure before 1 year of age may have been a precipitating factor for developing zoster. He also had a sustained closed head injury prior to admission, which led us to suspect that a systemic immune response to the injury might have contributed to the onset of herpes zoster ophthalmicus and VZV meningoencephalitis.
The incidence and severity of herpes zoster and neurological complications are associated with decreased cell-mediated immunity to VZV [6]. Although the VZV-specific T cell counts could not be determined in this patient, we believe that quantitative decreases in CD4+ T-lymphocytes limit the adaptive immunity to VZV, thereby increasing the risk of meningoencephalitis.
This patient did not show pleocytosis in the CSF. Only 38% of patients with herpes zoster have elevated lymphocytes in their CSF [7]. A shorter period from the onset of meningitis to the clinical examination increases the likelihood of not detecting CSF pleocytosis. For a more reliable diagnosis of meningoencephalitis, performing follow-up CSF analysis a few days later to check for pleocytosis may be helpful. In our patient, a follow-up CSF analysis was not performed due to progressive lethargy, which is a contraindication for lumbar puncture.
As the patient's consciousness declined, electroencephalography (EEG) would have also been indicated to investigate for encephalopathy, but EEG was not performed because his shingles encompassed the entire right forehead, rendering an examination impossible (Fig. 1B and C). Finally, a positive PCR test result for VZV in CSF with a typical vesicular rash and progressive lethargic consciousness led us to conclusively diagnose aseptic zoster encephalitis, although there was no pleocytosis in the CSF study and EEG was not possible.
Children with VZV encephalitis regularly undergo central nervous system imaging studies that typically yield normal results. However, magnetic resonance imaging may occasionally show edema or white matter lesions that suggest acute demyelinating encephalomyelitis. Neuroimaging studies did not show significant findings in our patient.
Although treatment for zoster meningitis in immunocompetent children has not been established, zoster meningitis should be treated promptly and aggressively to prevent complicated zoster meningoencephalitis. To treat complicated zoster, acyclovir (10 mg/kg/dose or 500 mg/m2/dose) every 8 hours is recommended for 10 to 14 days [8]. The patient was administered intravenous acyclovir at a dose of 30 mg/kg/day. However, it was insufficient to induce resolution of the skin rash. His cutaneous manifestations improved after increasing the dose of acyclovir to 1,500 mg/m2/day (equivalent to 60 mg/kg/day); the patient received acyclovir for a total of 24 days.
Herpes zoster may occur at any time after varicella infection or vaccination [9]. Pediatric zoster is typically caused by reactivation after natural infection with VZV. However, among vaccinated children, up to half of cases occur after immunization [9]. We could not evaluate whether our patient’s zoster resulted from the reactivation of wild-type VZV or vaccine-strain VZV. Vaccine-strain herpes zoster favors the lumbar and cervical dermatomes, possibly reflecting vaccinations in the thigh and upper arm. Most wild-type herpes zoster infections are thoracic and typically involve older children [9]. Since several cases of vaccine-strain herpes zoster in the trigeminal nerve area have been reported, the possibility of reactivation due to vaccination cannot be ruled out, even in this case [10].
Zoster meningoencephalitis is very rare among young children. Even if pleocytosis is absent in the initial CSF test or no typical cutaneous vesicles are observed in previously healthy young children, zoster meningoencephalitis should be considered as a differential diagnosis of aseptic meningitis in immunocompetent young children.
This study was approved by the Institutional Review Board of CHA Bundang Medical Center (approval number: 2021-03-013). Written informed consent by the patients was waived due to a retrospective nature of our study.
Notes
No potential conflict of interest relevant to this article was reported.
Author contribution
Conceptualization: HJ, SR, and KYC. Data curation: HJ and KYC. Formal analysis: HJ, SR, and KYC. Funding acquisition: KYC. Methodology: HJ and KYC. Project administration: HJ and KYC. Visualization: HJ. Writing-original draft: HJ. Writing-review & editing: HJ, SR, and KYC.

