|
 |
- Search
| Ann Child Neurol > Volume 29(2); 2021 > Article |
|
Abstract
Purpose
This study evaluated the clinical and laboratory characteristics of infants ≤90 days old with meningitis who presented to the hospital with a fever. We also investigated whether initial C-reactive protein levels and white blood cell counts were reliable predictors of bacterial meningitis.
Methods
The medical records of 1,151 infants aged ≤90 days who visited our hospital with a fever between October 2009 and October 2019 were retrospectively evaluated.
Results
Of the 1,151 patients, 274 (23.8%) had meningitis (bacterial, n=7; viral, n=206; pleocytosis in the cerebrospinal fluid, n=136). Thirty-seven viral meningitis patients (18.0%) had a positive polymerase chain reaction result without pleocytosis in the cerebrospinal fluid. The patients without pleocytosis were significantly younger. Among the patients with only pleocytosis, 46 had a urinary tract infection, 22 had other viral infections, and the etiology was unknown in 68. Among patients with urinary tract infections, infants without pleocytosis were younger than those with pleocytosis. Low white blood cell counts (<5,000/mm3) were more frequently found in bacterial meningitis patients (n=7) than in viral meningitis patients. Furthermore, there were normal C-reactive protein levels (42.9%) and no pleocytosis (20%) in some cases of bacterial meningitis.
Conclusion
Our findings show that meningitis is not uncommon among infants ≤90 days old who were brought to the hospital with complaints of fever. Furthermore, younger patients may not have cerebrospinal fluid pleocytosis, even if they have bacterial meningitis. Therefore, the patient’s condition should be monitored closely and, if necessary, a re-examination should be considered.
Fever is the most common symptom indicative of serious infections among young infants, including neonates [1]. Although most febrile young infants have simple viral infections [2], bacterial infections, such as urinary tract infections (UTIs), meningitis, and bacteremia, are not uncommon.
Central nervous system infections, such as bacterial or viral meningitis, frequently occur in young infants due to immature humoral and cellular immunity. Depending on the age at diagnosis, type of identified organism(s), and delay in treatment, central nervous system infections can lead to several acute comorbidities, severe complications, and long-term disabilities, ranging from hearing loss to a permanent motor or cognitive impairment [3]. An early diagnosis of meningitis in younger infants is essential for correct treatment so as to reduce mortality and complications.
Clinical signs and symptoms of central nervous system infections are often nonspecific in young infants. These symptoms include fever, hypothermia, food retention, skin lesions, irritability, or general malaise [4,5]. Therefore, blood and cerebrospinal fluid (CSF) analyses are performed to diagnose or rule out severe central nervous system infections. A lumbar puncture (LP) with an analysis of the CSF profile might help the clinician differentiate between the presence or absence of meningitis and between bacterial or viral meningitis. CSF white blood cell (WBC) counts >1,000 cells/mm3 suggest bacterial meningitis [6]. The presence of CSF pleocytosis is usually diagnosed with meningitis. However, the absence of CSF pleocytosis in younger infants with enteroviral meningitis has been previously reported [7-9].
In addition, serum C-reactive protein (CRP) levels, WBC count, and procalcitonin are commonly used inflammatory biomarkers in the blood. However, there is no consensus regarding their ability to distinguish between bacterial and viral meningitis [10].
In this study, we aimed to describe the biochemical characteristics of CSF, blood, and urine in febrile infants ≤90 days old with meningitis. Furthermore, we compared the characteristics of the laboratory results in patients with meningitis after dividing them into two groups based on the presence or absence of pleocytosis in the CSF.
The medical records of 1,172 patients of age less than 90 days who visited Inje University Sanggye Paik Hospital with a complaint of fever between October 2009 and October 2019 were retrospectively evaluated. The study protocol was approved by the Institutional Review Board of Inje University Sanggye Paik Hospital (2020-03-015). The requirement for informed consent was waived due to the retrospective nature of the study.
Patients <35 weeks gestational age (n=7) and those with underlying conditions (n=14), including Cornelia de Lange syndrome (n=1), cerebral infarction (n=2), periventricular leukomalacia (n=1), Prader-Willi syndrome (n=1), congenital cytomegalovirus infection (n=1), and congenital hypothyroidism (n=1), were excluded. Data regarding age, gestational age, sex, clinical characteristics, diagnosis, and all laboratory profiles, including blood, CSF, urine, and sputum were collected from all the patients.
For all included patients, all blood laboratory tests including initial WBC and CRP level checks were performed immediately after visiting the hospital. The peak CRP was defined as the highest numerical value by comparing all tests performed during hospitalization, including the initial CRP.
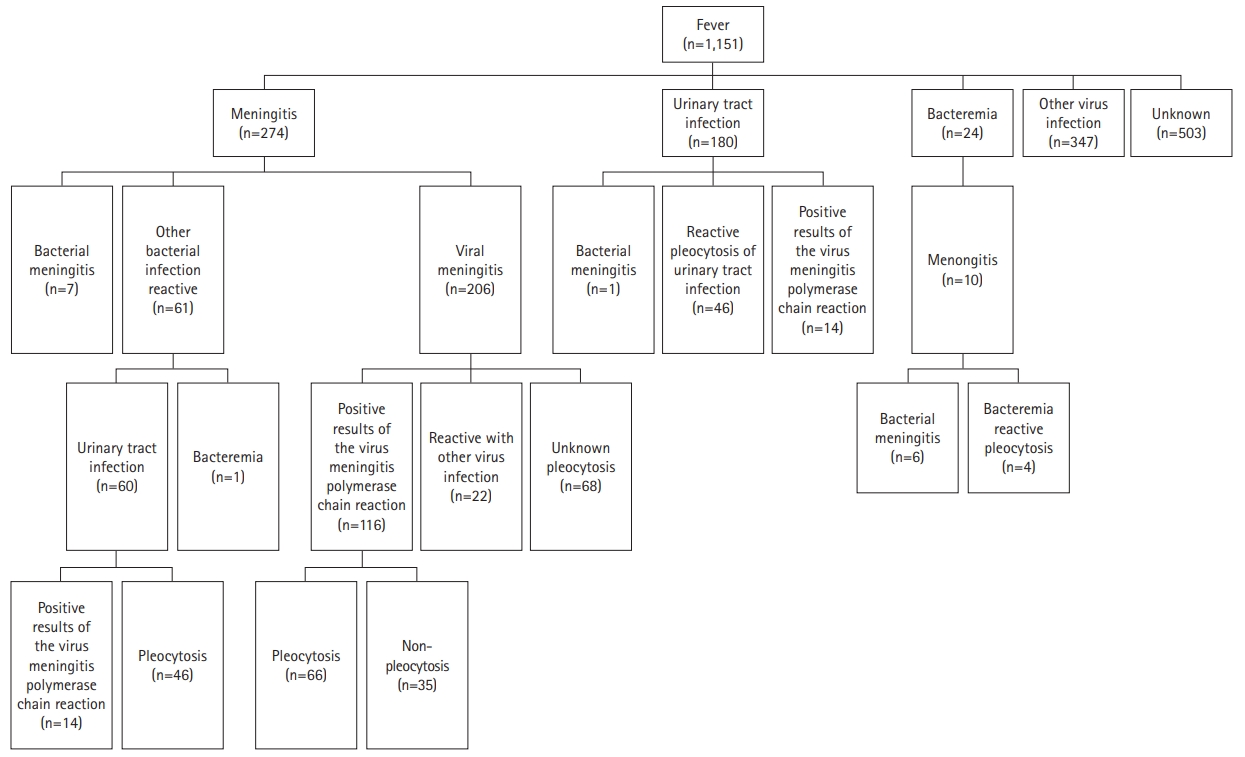
For all the included patients (n=1,151), LP is usually performed after the initial blood test following which the platelet level is confirmed. In our study, LP was performed on 867 patients (74.3%). Of those 867 patients, 281 did not undergo CSF analysis due to traumatic tap or insufficient sample quantity, and one patient had a positive result for virus polymerase chain reaction (PCR) of enterovirus. Patients with traumatic LP were excluded from the CSF analysis. The detailed results are described in Fig. 1.
All patients who received the LP were tested for bacterial culture and underwent real-time multiplex PCR (Seegene Inc., Seoul, Korea) for six types of viruses—cytomegalovirus, human herpesvirus 6, Epstein-Barr virus, herpes simplex viruses 1 and 2, and varicella-zoster virus—or Reverse Transcriptase PCR for enteroviruses (Seegene Inc.).
Bacterial meningitis was defined as bacterial growth in the CSF. Viral meningitis was defined as either a positive PCR result or patients with age-adjusted CSF pleocytosis with a negative CSF culture or PCR results for a virus without UTIs or bacteremia. Coinfections were defined as meningitis accompanied by bacteremia, UTI, or other viral infections.
CSF pleocytosis was defined as a CSF WBC count >22 WBCs/mm3 if the patient was <4 weeks old; >15 WBCs/mm3 if the patient was between 4 and 7 weeks of age; and >5 WBCs/mm3 if the patient was ≥8 weeks old. UTI was diagnosed as a urine culture (collected by urethral catheterization) with more than 100,000 colony-forming units/mL [11]. The growth of bacteria that are not commonly considered as pathogens (Staphylococcus epidermidis or coagulase-negative Staphylococcus) was classified as a priori as contamination [12].
Patients with various types of meningitis were compared and analyzed based on the presence or absence of pleocytosis in the CSF.
Continuous variables, including age and CRP, are expressed as the mean±standard deviation and were compared using t-tests. Categorical variables, including sex and age group, were compared using a chi-square test or Fisher’s exact test. The comparison between the viral meningitis group and bacterial meningitis was performed a nonparametric test, Mann-Whitney U test. All statistical analyses were performed using IBM SPSS Statistics for Windows version 25.0 (IBM Corp., Armonk, NY, USA). Statistical significance was defined as a P<0.05.
During the study period, 1,151 infants (630 [54.74%] males and 521 [45.26%] females) were included. The cause of infection was identified in 724 (432 [59.67%] males and 292 [40.33%] females). LP was performed on 867 patients: meningitis (n=274, 23.8%); UTI (n=180); bacteremia (n=24); and other viral infections (n=347). The remaining 503 patients with unexplained fever were suspected of having a viral infection and they recovered without complications (Fig. 2).
Of the 274 meningitis patients, 158 (57.7%) were males and 116 (42.3%) were females. The mean age at symptom onset was 52.85±23.42 days (range, 1 to 90). Fifty-nine (21.5%) infants were ≤30 days old, 94 (34.3%) were 31 to 60 days old, and 121 (44.2%) were 61 to 90 days old. Seven patients had bacterial meningitis (Group B Streptococcus [GBS], n=5; Escherichia coli, n=2), 206 had viral meningitis, and 136 had only pleocytosis in the CSF (Table 1). Of the 130 patients with viral meningitis who had a positive viral PCR test, 37 (17.9%) had a positive PCR result without pleocytosis in the CSF. Of the 136 patients with meningitis with only pleocytosis, 46 had a UTI, 22 were reactive in other viral infections, and 68 were of an unknown etiology. The detailed information is described in Fig. 2.
Among the patients with bacterial meningitis, except for two patients with traumatic tap who could not be included in the analysis, 80% (4/5) had pleocytosis and 20% (1/5) had non-pleocytosis CSF. One patient who had non-pleocytosis with the initial WBC count and initial CRP in a normal range was later diagnosed with GBS meningitis. In addition, another patient with traumatic tap had CSF non-pleocytosis (CSF red blood cell count, >20,000/mm3; CSF WBC, 3/mm3). Therefore, 28.6% (2/7) of bacterial meningitis patients had non-pleocytosis.
A comparison of the bacteremia patients with and without pleocytosis in the CSF revealed that a significantly large proportion of patients without pleocytosis underwent LP <24 hours after the onset of fever (10/11, 90.9%; chi-square test, P=0.046).
Of the 191 children diagnosed with viral meningitis without traumatic tap, 81.7% (156/191) had CSF pleocytosis and 18.3% (35/191) had CSF non‐pleocytosis (Table 2). The comparison of viral meningitis with and without pleocytosis showed no significant difference in CRP (Table 2). However, patients without pleocytosis were significantly younger (39.20±22.64 days vs. 54.19±22.16 days, t-test P<0.05) and had lower WBC counts (12,757±5,358.75/mm3 vs. 9,134±3,507.66/mm3, t-test P<0.05). A greater proportion of patients without pleocytosis underwent LP <24 hours after the onset of fever (chi-square test, P=0.006) than patients with pleocytosis (Table 2). The multivariate logistic regression analysis showed that younger age, shorter interval from fever onset to LP, lower WBC in the peripheral blood, lower CSF protein, and higher CSF glucose were independent predictors of CSF non-pleocytosis in viral meningitis in infants ≤90 days old (Table 2).
There were no patients with bad prognosis from viral meningitis. However, one of the patients with bacterial meningitis died and one suffered from subdural empyema, but is developing normally without any sequelae.
Among the patients with UTIs, 7.8% had coinfection with PCR-positive viral meningitis, 25.6% had reactive pleocytosis in the CSF, and one patient (0.5%) had a co-infection with bacterial meningitis. A comparison of the patients with UTI with and without pleocytosis in the CSF revealed that younger patients had a low rate of pleocytosis occurrence (56.23±20.41 days vs. 67.61±13.59 days, t-test P<0.05). A greater proportion of patients without pleocytosis underwent LP <24 hours after the onset of fever (chi-square test, P=0.001) compared to patients with pleocytosis. This further indicates that younger age and early examination correlate with the absence of pleocytosis.
When comparing bacterial meningitis with viral meningitis, there were more cases of WBC count <5,000/mm3 in patients with bacterial meningitis (chi-square test, P<0.05) (Table 3). Moreover, the initial CRP levels (0.50 mg/dL [0.3-11.3] vs. 1.00 mg/dL [0.3-13.9], respectively; Mann-Whitney U test, P=0.024) (Table 3) and peak CRP levels (0.60 mg/dL [0.3-14.7] vs. 12.90 mg/dL [6.8-26.7], respectively; Mann-Whitney U test, P=0.000) (Table 3) were significantly different. Initial WBC counts <5,000/mm3 were more frequent in bacterial meningitis cases but CRP levels were significantly different between viral and bacterial meningitis. However, among bacterial meningitis (n=7), three patients (42.9%) had normal CRP level initially.
It is challenging to differentiate between severe infection and benign viral infections among infants aged ≤90 days reporting with a fever. Of the severe infections in young infants, central nervous system infections are the most serious as they may lead to long-term morbidities. However, the most common infection of the central nervous system is aseptic meningitis. In a study of young infants with acute meningitis, almost 80% of infants were found to have aseptic meningitis [3]. In our study, 274 patients (31.6%) were diagnosed with meningitis. Of these, seven had bacterial meningitis, 206 had viral meningitis, and 68 had pleocytosis in the CSF with an unknown cause (Table 1 and Fig. 1).
Of the 191 children diagnosed with viral meningitis without traumatic tap, 18.3% had CSF non‐pleocytosis. In particular, infants with CSF non‐pleocytosis were younger than those with CSF pleocytosis, suggesting a negative correlation with age (Table 2). Univariate analysis showed that a shorter interval from symptom onset to LP, lower CSF glucose, and lower WBC count in the peripheral blood were significantly associated with viral meningitis with CSF non‐pleocytosis (Table 3). In our study, 28.6% of bacterial meningitis patients did not have pleocytosis; however, the number of patients was too small to compare pleocytosis and no pleocytosis. Thus, it might be challenging to differentiate between bacterial and viral meningitis based on initial CSF analysis, as there might be overlaps in the CSF white cell count.
Previous studies also showed some cases of enteroviral meningitis without pleocytosis [7]. The proportion of patients with CSF non‐pleocytosis in the case of enteroviral meningitis was approximately 30% of infants <2 months of age in New Zealand [13] and in Missouri (USA) [14], and 31% in Pennsylvania (USA) [15]. In Korea, approximately 28% of infants <3 months of age [16], 63.3% of neonates, and 38.3% of young infants aged 29 to 56 days [8] had CSF non-pleocytosis in enteroviral meningitis. Mulford et al. [17] reported that 30% of infants <2 months of age with enteroviral meningitis did not have pleocytosis. Although studies published so far have focused upon enteroviral meningitis and CSF pleocytosis, this study shows that various types of meningitis and not only meningitis caused by an enterovirus, may be related to meningitis without CSF pleocytosis in younger patients who were ≤90 days old.
The mechanism of meningitis with non-pleocytosis in infants is still unclear. Seiden et al. [9] reported that CSF non‐pleocytosis is associated with lower peripheral WBC counts and suggested this may result from a lack of sufficient cellular response to infections.
In our study, the mean time interval from onset to LP was significantly shorter in viral meningitis without pleocytosis. These results suggest that sufficient time may be needed for CSF pleocytosis to develop. In other words, in the early stages of central nervous system infections, results may indicate non-pleocytosis if sufficient time has not passed to allow pleocytosis to develop [8,18,19].
Our study showed that 7.8% of UTI patients had coinfection with PCR-positive viral meningitis, 25.6% had reactive pleocytosis in CSF, and one patient (0.5%) had bacterial meningitis (E. coli). Previous studies have shown that sterile CSF pleocytosis is not rare in infants with a UTI undergoing LP (18% to 29% of patients); however, UTIs are rarely associated with bacterial meningitis [20,21]. It is thought that pleocytosis is a type of inflammatory response to UTI and is the result of undetected viral infections [22,23]. In this study, among the patients with UTIs, infants without pleocytosis were younger than those with pleocytosis. It is unknown whether sterile CSF pleocytosis in UTIs is significant and further research on this topic is warranted.
Meningitis may not always be accompanied by pleocytosis. Pleocytosis can be related to age, the interval from symptom onset to LP, and peripheral WBC counts. A comparison between viral meningitis and bacterial meningitis revealed that there were more cases of WBC count <5,000/mm3 in patients with bacterial meningitis (chi-square test, P<0.05) (Table 3). This suggests that bacterial meningitis may present with relatively low WBC counts. Furthermore, CSF non-pleocytosis may also be present. Although the initial CRP levels were significantly different between viral and bacterial meningitis (0.50 mg/dL [0.3-11.3] vs. 1.00 mg/dL [0.3-13.9], respectively; Mann-Whitney U test, P=0.024) (Table 3), initial CRP levels were normal in three cases of seven bacterial meningitis (3/7, 42.9%). Together, these results suggest that the distinction between viral and bacterial meningitis may be potentially difficult in the early stages of disease onset, leading to delayed diagnosis. Therefore, the patient’s condition should be monitored closely and, if necessary, re-examinations should be considered.
Excluding herpes simplex virus infections, most viral meningitis is known to be non-fatal. However, some cases of enterovirus, human herpesvirus 6 infections, and parechovirus [6,24,25] may be accompanied by CSF non-pleocytosis and may cause severe encephalitis, which can be fatal or lead to further complications. Therefore, PCR tests to identify the specific virus that caused meningitis are needed; these tests have the added benefits of reducing the need for further investigations, the number of medications prescribed, and the length of hospital stay.
This study has some limitations. As our work is retrospective in nature, a generalization of the results may not be appropriate. We did not obtain data of CSF examinations in all the included infants. Therefore, further prospective studies are necessary.
In conclusion, we showed that meningitis is not uncommon among infants ≤90 days old who have come to the hospital with a fever. Furthermore, younger age may be negatively correlated with CSF pleocytosis in meningitis. Our results indicate initial CRP levels may not be reliable predictors of bacterial meningitis and that initial leukopenia may be more dependable. Analysis of CSF or inflammatory markers in the peripheral blood is insufficient to diagnose meningitis and to also differentiate between viral and bacterial source, and therefore, PCR tests for various viruses and bacteria may be necessary. Meningitis patients <90 days old have non-specific symptoms and signs and often do not have CSF pleocytosis. It is crucial to closely monitor the patient's condition by analyzing whether the laboratory findings and clinical symptoms are consistent.
Notes
Author contribution
Conceptualization: SJY. Data curation: KUC. Formal analysis: KUC. Writing-original draft: KUC. Writing-review & editing: KUC and SJY.
Acknowledgments
We would like to thank Ho Hyeong Jo for his time and useful comments. This work was supported by the 2019 Inje University research grant.
Table 1.
Clinical characteristics of febrile infants with meningitis (n=274)
| Characteristic | Value |
|---|---|
| All patients | 274 |
| Age (day) | 52.85±23.42 (1-90) |
| Age group (day) | |
| 0-30 | 59 (21.5) |
| 31-60 | 94 (34.3) |
| 61-90 | 121 (44.2) |
| Sex, male:female | 158 (57.7):116 (42.3) |
| Bacterial meningitisa | 7 |
| Group B Streptococcus (Streptococcus agalactiae) | 5 |
| Escherichia coli | 2 |
| Viral meningitis with virus PCR positive in CSFb | 130 |
| Type of virus in cases with positive PCR in CSF | |
| Human herpesvirus-6 meningitis | 1 |
| Enteroviral meningitis | 129 |
| Type of viral meningitis excluding patients without CSF analysis | 113 |
| Viral PCR (+) in CSF with pleocytosis | 76 |
| Viral PCR (+) in CSF without pleocytosis | 37 |
| Traumatic tap | 16 |
| Viral PCR (+) without CSF analysis | 1 |
| Only pleocytosis in CSF | 136 |
| Unknown pleocytosis | 68 |
| Reactive in urinary tract infection | 46 |
| Reactive in other viral infection | 22 |
| Reactive in bacteremia | 1 |
| Urinary tract infection with viral meningitis (virus PCR positive) | 14 |
Table 2.
Comparison of viral meningitis with and without pleocytosis in cerebrospinal fluid
| Variable | With pleocytosis (n=156) | Without pleocytosis (n=35) | P value | Multivariate analysis |
|---|---|---|---|---|
| Male | 84 | 12 | 0.04a | P>0.05 |
| Female | 72 | 23 | ||
| Age (day) | 54.18±22.23 | 39.20±22.64 | 0.00a | P=0.00a |
| Exp(B)=1.07 | ||||
| (1.04-1.10) | ||||
| Age group (day) | ||||
| <60 | 86 | 29 | 0.00a | |
| >60 | 70 | 6 | ||
| Time interval from onset to LP (day) | 0.84±1.09 | 0.43±0.82 | 0.04a | P>0.05 |
| <24 hours | 67 (42.95) | 24 (68.57) | 0.01a | |
| CSF WBC (/mm3) | 182.94±277.05 | 3.94±4.26 | 0.00a | |
| CSF protein (mg/dL) | 78.01±34.15 | 70.27±27.05 | >0.05 | P=0.00a |
| Exp(B)=1.04 | ||||
| (1.01-1.06) | ||||
| CSF glucose (mg/dL) | 49.39±12.56 | 55.11±11.02 | 0.01a | P=0.03a |
| Exp(B)=0.96 | ||||
| (0.93-0.10) | ||||
| Peripheral blood | ||||
| WBC (/mm3) | ||||
| <5,000 | 6 | 3 | >0.05 | |
| >15,000 | 49 | 3 | 0.01a | |
| Mean | 12,792.56±5,357.56 | 9,134.57±3,507.66 | 0.00a | P=0.01a |
| Exp(B)=1.00 | ||||
| (1.00-1.00) | ||||
| Initial CRP (mg/dL) | 1.10±1.64 | 1.06±1.02 | >0.05 | P>0.05 |
| Peak CRP (mg/dL) | 1.40±2.61 | 1.45±1.46 | >0.05 | P>0.05 |
Table 3.
Comparison between viral meningitis and bacterial meningitis patients
| Variable | Viral meningitisa (n=206) | Bacterial meningitis (n=7) | Comparison P value |
|---|---|---|---|
| Age (day) | 53.00 (1-90) | 27.00 (2-74) | 0.03a |
| Sex | >0.05 | ||
| Male | 103 | 5 | |
| Female | 103 | 2 | |
| Time interval from onset to LP (day) | 1.00 (0-7) | 0.00 (0-1) | >0.05 |
| <24 hours | 100 (48.54) | 6 (85.71) | >0.05 |
| CSF WBC (/mm3) | 40.00 (0-1,360) | 230.00 (2-1,970) | >0.05 |
| CSF protein (mg/dL) | 74.65 (19-501.2) | 111.40 (60.3-566.6) | >0.05 |
| CSF glucose (mg/dL) | 49.00 (29-127) | 52.30 (7.6-85.7) | >0.05 |
| Initial white blood cell count | |||
| <5,000/mm3 | 10 (4.83) | 2 (28.57) | 0.01a |
| >15,000/mm3 | 53 (25.60) | 2 (28.57) | >0.05 |
| Median | 11,125 (1,540-27,120) | 10,210 (1,790-31,810) | >0.05 |
| Initial C-reactive protein | 0.5 (0.3-11.3) | 1.0 (0.3-13.9) | 0.02a |
| Peak C-reactive protein | 0.6 (0.3-14.7) | 12.9 (6.8-26.7) | 0.00a |
References
1. Baskin MN. The prevalence of serious bacterial infections by age in febrile infants during the first 3 months of life. Pediatr Ann 1993;22:462-6.


2. Sharp J, Harrison CJ, Puckett K, Selvaraju SB, Penaranda S, Nix WA, et al. Characteristics of young infants in whom human parechovirus, enterovirus or neither were detected in cerebrospinal fluid during sepsis evaluations. Pediatr Infect Dis J 2013;32:213-6.



3. Abdelmaguid N, Seleem WS, Soliman AT, Mohamed RS, Elgharbawy FM, Yassin H, et al. Clinical presentations, laboratory analysis and linear growth in 50 neonates and young infants with acute meningitis: one year experience of a single center in Qatar. Mediterr J Hematol Infect Dis 2019;11:e2019028.



4. Barichello T, Fagundes GD, Generoso JS, Elias SG, Simoes LR, Teixeira AL. Pathophysiology of neonatal acute bacterial meningitis. J Med Microbiol 2013;62:1781-9.


5. Ku LC, Boggess KA, Cohen-Wolkowiez M. Bacterial meningitis in infants. Clin Perinatol 2015;42:29-45.


6. Hysinger EB, Mainthia R, Fleming A. Enterovirus meningitis with marked pleocytosis. Hosp Pediatr 2012;2:173-6.


7. Yun KW, Choi EH, Cheon DS, Lee J, Choi CW, Hwang H, et al. Enteroviral meningitis without pleocytosis in children. Arch Dis Child 2012;97:874-8.


8. Song JY, Nam SO, Kim YA, Kim KM, Lyu SY, Ko A, et al. Cerebrospinal fluid non-pleocytosis in pediatric enteroviral meningitis: large-scale review. Pediatr Int 2018;60:855-61.


9. Seiden JA, Zorc JJ, Hodinka RL, Shah SS. Lack of cerebrospinal fluid pleocytosis in young infants with enterovirus infections of the central nervous system. Pediatr Emerg Care 2010;26:77-81.


10. American College of Emergency Physicians Clinical Policies Committee; American College of Emergency Physicians Clinical Policies Subcommittee on Pediatric Fever. Clinical policy for children younger than three years presenting to the emergency department with fever. Ann Emerg Med 2003;42:530-45.


11. Bonadio WA, Stanco L, Bruce R, Barry D, Smith D. Reference values of normal cerebrospinal fluid composition in infants ages 0 to 8 weeks. Pediatr Infect Dis J 1992;11:589-91.


12. Mahajan P, Kuppermann N, Mejias A, Suarez N, Chaussabel D, Casper TC, et al. Association of RNA biosignatures with bacterial infections in febrile infants aged 60 days or younger. JAMA 2016;316:846-57.



13. Mazor SS, McNulty JE, Roosevelt GE. Interpretation of traumatic lumbar punctures: who can go home? Pediatrics 2003;111:525-8.



14. Kost CB, Rogers B, Oberste MS, Robinson C, Eaves BL, Leos K, et al. Multicenter beta trial of the GeneXpert enterovirus assay. J Clin Microbiol 2007;45:1081-6.



15. Casas I, Palacios GF, Trallero G, Cisterna D, Freire MC, Tenorio A. Molecular characterization of human enteroviruses in clinical samples: comparison between VP2, VP1, and RNA polymerase regions using RT nested PCR assays and direct sequencing of products. J Med Virol 2001;65:138-48.


16. Elmore JG, Horwitz RI, Quagliarello VJ. Acute meningitis with a negative Gram's stain: clinical and management outcomes in 171 episodes. Am J Med 1996;100:78-84.


17. Mulford WS, Buller RS, Arens MQ, Storch GA. Correlation of cerebrospinal fluid (CSF) cell counts and elevated CSF protein levels with enterovirus reverse transcription-PCR results in pediatric and adult patients. J Clin Microbiol 2004;42:4199-203.



18. Garges HP, Moody MA, Cotten CM, Smith PB, Tiffany KF, Lenfestey R, et al. Neonatal meningitis: what is the correlation among cerebrospinal fluid cultures, blood cultures, and cerebrospinal fluid parameters? Pediatrics 2006;117:1094-100.


19. Greenberg RG, Benjamin DK Jr, Cohen-Wolkowiez M, Clark RH, Cotten CM, Laughon M, et al. Repeat lumbar punctures in infants with meningitis in the neonatal intensive care unit. J Perinatol 2011;31:425-9.


20. Thomson J, Cruz AT, Nigrovic LE, Freedman SB, Garro AC, Ishimine PT, et al. Concomitant bacterial meningitis in infants with urinary tract infection. Pediatr Infect Dis J 2017;36:908-10.


21. Schnadower D, Kuppermann N, Macias CG, Freedman SB, Baskin MN, Ishimine P, et al. Sterile cerebrospinal fluid pleocytosis in young febrile infants with urinary tract infections. Arch Pediatr Adolesc Med 2011;165:635-41.


22. Doby EH, Stockmann C, Korgenski EK, Blaschke AJ, Byington CL. Cerebrospinal fluid pleocytosis in febrile infants 1-90 days with urinary tract infection. Pediatr Infect Dis J 2013;32:1024-6.



23. Adler-Shohet FC, Cheung MM, Hill M, Lieberman JM. Aseptic meningitis in infants younger than six months of age hospitalized with urinary tract infections. Pediatr Infect Dis J 2003;22:1039-42.


- TOOLS
-
METRICS

-
- 0 Crossref
- Scopus
- 4,224 View
- 112 Download
- Related articles in Ann Child Neurol
-
Characteristics of Meningitis with or without Enterovirus2018 December;26(4)
Characteristics of Sleep Disorders in Childhood and Adolescence.2007 November;15(2)