 |
 |
- Search
| Ann Child Neurol > Volume 27(2); 2019 > Article |
|
Abstract
Purpose
Acquired epileptic aphasia (AEA) accompanied by electroencephalogram (EEG) abnormality is a rare disease; therefore, there are few studies investigating the prognostic factors and treatment efficacy. We aimed to determine the therapeutic effects and prognostic factors for clinical seizure and neuropsychological function in acquired aphasia patients.
Methods
We retrospectively studied cases of AEA diagnosed at Severance Children's Hospital from January 2013 to October 2017. We evaluated the efficacy of antiepileptic drugs, steroids, and ketogenic diets (KD) in treating acquired aphasia. The EEG patterns and prognostic factors were predicted by the background EEG and frequency of spike and wave during sleep (SWS).
Results
The study analyzed 20 patients, 11 male and 9 female, with AEA. Aphasia most commonly occurred at 4 years of age, and clinical seizure was most likely to occur between 2 and 4 years of age and focal seizures were the most common seizure type. KD was shown to be the best treatment for clinical seizure in AEA patients. Patients with normal EEG background showed better responses to clinical seizure treatment and improvements in neuropsychological function.
Conclusion
KD and steroids generate the best therapeutic effects for clinical seizure in AEA patients. Improvements in neuropsychological function in AEA patients may be related to the EEG background and the SWS patterns. Additionally, the results suggest that the response of clinical seizure to antiepileptic drugs may also be related to the EEG background. However, the current study had some limitations and further research is needed.
Acquired epileptic aphasia is a sudden or progressive language impairment with an abnormality in the electroencephalogram (EEG) [1-3]. Some researchers have reported that epileptiform EEG discharges can affect not only language impairment but also neurocognitive function [3,4]. Language impairment with an abnormality in the EEG is observed in some epilepsy syndrome, such as electrical status epilepticus during sleep (ESES), continuous spike and wave during slow sleep (CSWS), and Landau-Kleffner syndrome (LKS) [3]. In much of the previous literature, CSWS and ESES were used interchangeably and LKS considered a subtype of CSWS [2-5].
Acquired epileptic aphasia is commonly referred to as LKS, which was first introduced in 1957 [1]. It usually shows auditory agnosia with focal or multifocal spikes or spike and wave discharges on the EEG which are continuous or nearly continuous during sleep [5].
CSWS shows general developmental difficulties including linguistic problems, and more than 85% of continuous spikes and waves in the EEG are seen during sleep. Usually, these are seen as bilateral and symmetric, but it could be shown asymmetric or focal patterns [6-12]. CSWS shows premorbid central nervous system dysfunction whereas LKS rarely indicates such [5].
Acquired aphasia with clinical seizures is a rare disease, and the prognostic factor of treatment and the cognitive outcome were less studied. In this study, we investigated the prognosis of neuropsychological function and therapeutic effect of clinical seizures through EEG in patients who have a language impairment with clinical seizure without distinguishing between LKS and CSWS.
We retrospectively analyzed 20 patients (11 males, nine females) with acquired aphasia diagnosed at Severance Children's Hospital from January 2013 to October 2017. Patients with acquired aphasia were included in the study, with or without seizures. They were assessed via EEG for more than 4 hours, and this test was examined by the International 10 to 20 system. EEG background waves were divided into four patterns: normal, disorganized, generalized slowing (GSW), and focal slowing (FSW). The frequency of spike and wave during sleep (SWS) on the EEG was grouped according to frequency as ≥85% and 50% to 84%, and divided into generalized and focal types according to the pattern. The response to different treatments was analyzed using the antiepileptic drugs (AEDs), valproate (VPA), and levetiracetam (LEV), in addition to other treatments, namely steroid and ketogenic diets (KDs). Treatment effects were assessed by clinical seizure at each outpatient clinic. We consider a positive effect of treatment that the frequency of seizures was reduced by 50% or more, and continued these statuses for more than 6 months. If it did not meet the above criteria, the treatment option was added or changed. The evaluation of neuropsychological function was evaluated through evaluation tools such as Korean Wechsler Intelligence Scale for Children. The test was performed at the time of diagnosis and 3 to 6 months later, and the results were evaluated by comparing the two tests.
Patients were enrolled in the study if they showed language impairment irrespective of the type of clinical seizure, including cases with no specific findings on their brain magnetic resonance imaging.
We excluded epileptic encephalopathy such as Lennox-Gastaut syndrome, which may accompany other cognitive impairments. This study was approved by the Institutional Review Board (IRB) of Yonsei University (IRB no., 4-2016-0080). Informed consent was waived due to the retrospective nature of the study.
In our patient cohort, 11 patients (55%) were males and nine patients (45%) were females. Aphasia occurred at 2 to 9 years of age, with six patients (30%) occurring at the age of 4, three patients (15%) at the age of 3, and three patients (15%) at the age of 7. Most commonly, clinical seizure in acquired aphasia occurred at 2, 3, and 4 years of age. Their clinical seizures were focal motor seizures (12 patients, 60%), generalized motor seizures (six patients, 30%), and dyscognitive type seizures (two patients, 10%).
When the EEG background wave was analyzed, seven patients (35%) showed a normal background rhythm, whereas 11 patients (55%) showed a GSW background pattern. The most common EEG patterns during sleep were focal CSWS patterns ≥85%, which occurred in 10 patients (50%), and generalized CSWS patterns ≥85%, which occurred in six patients (20%).
The most effective treatment for seizure in acquired aphasia was KD, followed by steroid administration, with 100% and 90% treatment efficacy, respectively. We also analyzed the relationship between EEG background pattern and treatment efficacy. VPA was used in a total of 18 patients and showed a positive effect in 10 of these 18 patients (56%). In patients with GSW EEG background activity, five of 10 patients (50%) were effectively treated with VPA, and in patients with normal EEG background activity, five of six patients (83.3%) were effectively treated with VPA. LEV was used in 11 patients, of which seven patients were effectively treated by the drug. In patients with normal background EEG, LEV was effective at treating all three patients it was prescribed to. In patients with GSW EEG background, LEV was effective in treating four of six patients. Ten patients who did not respond to AEDs were treated with steroids, nine patients (90%) showed a response to treatment. Ten patients who had a recurrence after steroid therapy and did not respond to AEDs were treated with KD, 10 patients (100%) showed a response to treatment. Steroids were used in patients with normal EEG background (n=4), GSW (n=5), and FSW (n=1). In the case of GSW background rhythm, all five patients were effectively treated for the clinical seizure. Similarly, the one patient with FSW was also effectively handled for the clinical seizure by steroid administration. KD was used in patients with normal (n=3), disorganized (n=1), and GSW (n=6) EEG backgrounds, and was effective in treating all patients (Table 1). In the comparison of treatment effect on seizure and the frequency of SWS, VPA was an effective treatment in five of 10 patients (50%) with ≥85% focal SWS, whereas LEV was effective in one of three patients (33.3%) with ≥85% focal SWS. Steroids were an effective treatment for five of six patients (83.3%) and KD was effective in five of five patients (100%) with ≥85% focal SWS. In the case of ≥85% of generalized SWS, VPA was effective in four of six patients (66.7%), LEV was effective in three of five patients (60%), and both steroid and KD were effective in four of four patients (100%). The above contents are summarized in Table 1.
We compared neuropsychological function with EEG background activity and the frequency of SWS in acquired aphasia. Ten patients had normal neuropsychological function and 10 patients had delayed function. In patients with normal neuropsychological function, six patients (60%) showed normal EEG background and four patients (40%) showed GSW EEG background. In patients with normal EEG background, six patients (85.7%) showed normal neuropsychological function, and one patient (14.3%) showed delayed function. EEG background activity showed a GSW pattern in seven patients (70%) with delayed neuropsychological function. We also analyzed the correlation between pattern and frequency of SWS and neuropsychological function. There were 14 patients with focal abnormalities those included SWS ≥85% pattern and six patients with generalized SWS ≥85% on EEG. Seven patients (50%) with focal abnormality on EEG showed mild to moderate neuropsychological abnormality and seven patients (50%) with focal abnormality were normal neuropsychological function and three patients (50%) with generalized SWS ≥85% were normal cognitive function. However, one patient (17%) generalized SWS ≥85% on EEG showed severe neuropsychological abnormality (Fig. 1). This is the only patient with severe cognitive impairment in this study. In 10 patients with abnormal neuropsychological function, the functional improvement was seen in nine cases with non-generalized background rhythm or focal epileptiform discharges in EEG but neither GSW of EEG background nor generalized CSWS patients showed functional improvement (Table 2).
Our results show similar onset age of clinical symptoms to that reported by the existing literature [13]. In this study, the most common age of aphasia onset was 4 years, and the period of clinical seizure was most commonly 2 to 4 years of age. In our study, most patients with aphasia showed ≥85% CSWS, more commonly exhibiting focal CSWS patterns than the generalized CSWS pattern. The correlation between EEG background activity and treatment for clinical seizure was difficult to find. However, when patients with ≥85% focal CSWS and patients with ≥85% generalized CSWS were compared, it was found that overall the latter responded well to treatment. Among the treatments considered, steroid administration and KD was found to be most effective, consistent with previous studies [14-16]. The results also show, using two different AEDs, VPA and LEV, that patients with normal EEG background activity show greater treatment response, and those AEDs were more effective in patients with generalized CSWS patterns than focal patterns. The efficacy for clinical seizure patients with CSWS >85% on EEG and the generalized pattern seems to be better. However, the therapeutic effect of LEV for clinical seizure showed a better effect in less than 84% CSWS and simple focal abnormality of EEG epileptiform pattern. So, we propose keeping in mind the epileptiform pattern of EEG when treating clinical seizures with acquired aphasia.
At diagnosis, 50% of patients showed normal neuropsychological function, while 45% showed mild to moderate delayed function. Among the patients with normal neurological function, 60% showed normal EEG background, whereas 70% of patients with delayed neurological function showed GSW EEG background. Therefore, the results suggest that EEG background activity is related to neuropsychological function in patients with CSWS. Among the patients with delayed function, one patient showed no improvement following treatment. In the relationship between prognosis of neuropsychological function and EEG, we found that the generalized pattern on EEG got a poor outcome than other patterns and the patients with focal CSWS patterns were usually mildly delayed, whereas those with generalized CSWS patterns were severely delayed. Through these results, we could consider that the generalized pattern in the EEG affects cognitive function and would suggest that the EEG background pattern may help predict the prognosis of neuropsychological function.
We could consider that the EEG pattern will be helpful in the treatment of clinical seizure and prediction for neuropsychological prognosis in acquired aphasia. However, this study has a limitation due to a small number of patients identified in a single center. Additional studies will be needed in the future to elucidate this relationship further.
Acknowledgments
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare, Republic of Korea (grant number: HI18C0586).
Fig. 1.
The relationship between neuropsychological function and spikes and waves during sleep (SWS) patterns in the electroencephalogram (EEG). The X-axis represents the pattern of the EEG, and the Y-axis frequency represents the number of patient.
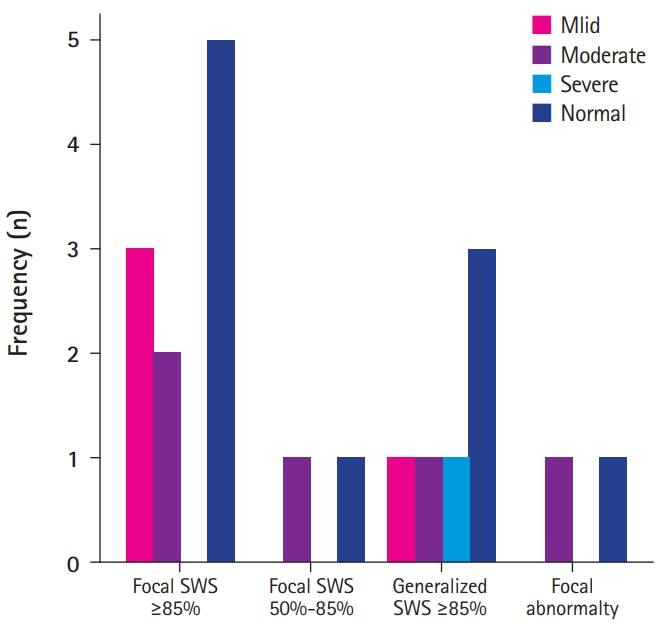
Table 1.
The effect of treatment for clinical seizure on EEG patterns
Table 2.
The relationship between neuropsychological functional outcome and EEG patterns
References
1. Landau WM, Kleffner FR. Syndrome of acquired aphasia with convulsive disorder in children. 1957. Neurology 1998;51:1241.


2. De Negri M. Electrical status epilepticus during sleep (ESES). Different clinical syndromes: towards a unifying view? Brain Dev 1997;19:447-51.


3. Overvliet GM, Besseling RM, Vles JS, Hofman PA, Backes WH, van Hall MH, et al. Nocturnal epileptiform EEG discharges, nocturnal epileptic seizures, and language impairments in children: review of the literature. Epilepsy Behav 2010;19:550-8.


4. Yung AW, Park YD, Cohen MJ, Garrison TN. Cognitive and behavioral problems in children with centrotemporal spikes. Pediatr Neurol 2000;23:391-5.


5. Galanopoulou AS, Bojko A, Lado F, Moshe SL. The spectrum of neuropsychiatric abnormalities associated with electrical status epilepticus in sleep. Brain Dev 2000;22:279-95.


6. Nickels K, Wirrell E. Electrical status epilepticus in sleep. Semin Pediatr Neurol 2008;15:50-60.


7. Sanchez Fernandez I, Loddenkemper T, Peters JM, Kothare SV. Electrical status epilepticus in sleep: clinical presentation and pathophysiology. Pediatr Neurol 2012;47:390-410.


8. Tassinari CA, Rubboli G, Volpi L, Meletti S, d'Orsi G, Franca M, et al. Encephalopathy with electrical status epilepticus during slow sleep or ESES syndrome including the acquired aphasia. Clin Neurophysiol 2000;111 Suppl 2:S94-102.


9. Bureau M, Genton P, Wolf P, Dravet C. Epileptic syndromes in infancy, childhood and adolescence. 5th ed. Montrouge: John Libbey Eurotext; 2012. p. 600.
10. Patry G, Lyagoubi S, Tassinari CA. Subclinical "electrical status epilepticus" induced by sleep in children. A clinical and electroencephalographic study of six cases. Arch Neurol 1971;24:242-52.


11. Van Hirtum-Das M, Licht EA, Koh S, Wu JY, Shields WD, Sankar R. Children with ESES: variability in the syndrome. Epilepsy Res 2006;70 Suppl 1:S248-58.


12. Fernandez IS, Peters J, Takeoka M, Rotenberg A, Prabhu S, Gregas M, et al. Patients with electrical status epilepticus in sleep share similar clinical features regardless of their focal or generalized sleep potentiation of epileptiform activity. J Child Neurol 2013;28:83-9.


13. Swaiman KF, Ashwal S, Ferriero DM, Schor NF, Finkel RS, Gropman AL, et al. Swaiman’s pediatric neurology: principles and practice. Edinburgh: Elsevier; 2017. p. 573-75.
14. Buzatu M, Bulteau C, Altuzarra C, Dulac O, Van Bogaert P. Corticosteroids as treatment of epileptic syndromes with continuous spike-waves during slow-wave sleep. Epilepsia 2009;50 Suppl 7:68-72.


- TOOLS







